Difference between revisions of "Sharing Laboratory Reports"
JohnMoehrke (talk | contribs) |
|||
| (34 intermediate revisions by 5 users not shown) | |||
| Line 1: | Line 1: | ||
| − | ''' Sharing Laboratory Reports (XD-LAB)''' | + | {| style="color: blue;" |
| + | | '''This profile is part of the Pathology and Laboratory Medicine (PaLM) domain, which merged the former AP and LAB domains since 2016, January 4th.''' | ||
| + | |} | ||
| + | ''' Sharing Laboratory Reports (XD-LAB)''' addresses the sharing of laboratory reports among a community of healthcare settings and care providers. | ||
| Line 7: | Line 10: | ||
==Summary== | ==Summary== | ||
| − | This Content Integration Profile describes a clinical laboratory report as an electronic document to be published towards a document sharing resource such as an Electronic Health Record (EHR) or | + | This Content Integration Profile describes a clinical laboratory report as an electronic document to be published towards a document sharing resource such as an Electronic Health Record (EHR) or a Personal Health Record (PHR) shared by a community of care providers, using one of the document sharing profiles defined in ITI-TF. Such an electronic document contains the set of releasable results produced by a clinical laboratory in fulfillment of one or more test Orders for a patient. <br/> |
| + | The report is both human-readable and importable in the consumer systems so as to consolidate their patient medical records. <br/> | ||
The scope of this profile covers all laboratory specialties except anatomic pathology. <br/> | The scope of this profile covers all laboratory specialties except anatomic pathology. <br/> | ||
| − | + | ||
| − | + | ||
<center>[[Image:ihe_lab_xd-lab_ip.jpg]]</center> | <center>[[Image:ihe_lab_xd-lab_ip.jpg]]</center> | ||
| Line 18: | Line 22: | ||
'''Facilitates Patient Care Coordination''' | '''Facilitates Patient Care Coordination''' | ||
| − | * Enables the caregiver to obtain at once the laboratory reports produced during previous episodes of care. | + | * Enables the caregiver to obtain at once the laboratory reports produced during previous episodes of care, which enhances continuity of care. |
| − | * | + | * Reduces over-ordering and over blood sampling from the patient by sharing the last laboratory reports among the various physicians attending the patient. |
* Enables the physician to consolidate their patient biological history by importing into their EHR the structured and coded data embedded in the electronic laboratory report. | * Enables the physician to consolidate their patient biological history by importing into their EHR the structured and coded data embedded in the electronic laboratory report. | ||
| + | * Allows a public health laboratory to share broadly its reports related to a suspected outbreak | ||
==Details== | ==Details== | ||
| − | The '''''Laboratory | + | The '''''Sharing Laboratory Reports (XD-LAB) Integration Profile''''' provides an electronic format for the laboratory report, to make this report: |
| − | This | + | * human-readable: displayed on screen or printed out, with a proper and clear layout and presentation. |
| − | The | + | * machine-readable: Each observation displayed in the report is also represented in the document as a set of structured and coded data that can be imported in the database of any consumer of the document.<br/> |
| − | * | + | This double capacity is achieved by leverageing the [Clinical Document Architecture Release 2] (CDAr2) standard from [[http://www.hl7.org HL7]]<br/> |
| − | * | + | The XD-LAB Integration Profile covers six major use cases: |
| − | * | + | * At discharge time, a hospital physician publishing a summary laboratory report with the most significant results obtained during the patient stay. |
| − | + | * The bio-medical scientist of a private laboratory publishes a report for a patient into the regional Patient Health Record. | |
| + | * An ambulatory physician shares a laboratory report obtained from a laboratory. | ||
| + | * A private or public laboratory publishes automatically all its reports in a shared document repository. | ||
| + | * A healthcare institution produces a cumulative report of all laboratory tests performed for the patient during the encounter. | ||
| + | * A public health laboratory shares its reports into a regional repository. | ||
| + | <br/> | ||
| + | |||
| + | The standard CDAr2 leveraged by XD-LAB enables to share reports embedding images and charts, which are faithfully rendered by a web browser supporting RFC 2397:<br/> | ||
| + | <center>[[Image:ihe_lab_xd-lab_example.jpg]]</center> | ||
| + | <br/> | ||
==Systems Affected== | ==Systems Affected== | ||
| Line 36: | Line 50: | ||
Systems involved in this profile are: | Systems involved in this profile are: | ||
| − | * | + | * A system owning a laboratory report and willing to publish it into a document sharing infrastructure. Depending upon the use cases, this can be the system of a clinical laboratory (LIS), the EHR of an ambulatory physician, the EMR of a care setting, the system of a public health laboratory. Each of these systems can play the role of Content Creator. |
| − | + | * The system operated by a professional caregiver who wants to view a laboratory report, and may want to import its structured and coded results into its own database. This system plays the role of Content Consumer. | |
| − | |||
| − | |||
'''Actors & Transactions:''' | '''Actors & Transactions:''' | ||
| − | [[Image: | + | <center>[[Image:xd-lab_actor_transaction.JPG]]</center> |
<br/> | <br/> | ||
| − | |||
| − | |||
| − | |||
==Specification== | ==Specification== | ||
| − | '''Profile Status:''' [[Comments| | + | '''Profile Status:''' [[Comments| Final Text]] |
| + | |||
| + | '''Documents:''' [https://www.ihe.net/resources/technical_frameworks/#PaLM IHE Laboratory Technical Framework:] | ||
| + | :* [https://www.ihe.net/uploadedFiles/Documents/PaLM/IHE_PaLM_TF_Vol1.pdf Vol. 1] - XD-LAB profile overview | ||
| + | :* [https://www.ihe.net/uploadedFiles/Documents/PaLM/IHE_PaLM_TF_Vol3.pdf Vol. 3] - Content | ||
| + | |||
| + | '''Computable specification:''' | ||
| + | :* [https://art-decor.ihe-europe.net/art-decor/decor-project--XDLAB- XD-LAB specified in Art-Decor on ihe-europe.net] - templates and value sets | ||
| − | |||
| − | |||
'''Underlying Standards:''' | '''Underlying Standards:''' | ||
| − | :* [http://www.hl7.org HL7] | + | :* [http://www.hl7.org HL7 CDA Release 2] |
| + | :* [http://www.regenstrief.org/medinformatics/loinc LOINC (Logical Observation Identifiers Names and Codes)] | ||
| + | :* [http://unitsofmeasure.org/trac The Unified Code for Units of Measure (UCUM)] | ||
| + | :* RFC 2397 | ||
==See Also== | ==See Also== | ||
| Line 62: | Line 79: | ||
'''Related Profiles''' | '''Related Profiles''' | ||
| − | * '''Integration Profiles | + | * '''Integration Profiles XD-LAB depends on''' |
| − | :* [[ | + | :* [[Cross Enterprise Document Sharing]] (XDS) or [[Cross_Enterprise_Document_Media_Interchange]] (XDM) or [[Cross_Enterprise_Document_Reliable_Interchange]] (XDR) from the ITI Technical Framework. Each of these three profiles provides an infrastructure for the sharing of medical documents between the creators and the consumers. |
| − | + | :* [[Audit Trail and Node Authentication]] (ATNA) from the ITI Technical Framework, to authenticate and audit the document-sharing transactions: Document storage and registration, document query and retrieval. | |
| − | + | :* [[Consistent Time]] (CT) from the ITI Technical Framework, to ensure accuracy of the time stamps in the shared documents and in the associated metadata. | |
| − | + | ||
| − | :* [[ | ||
| − | :* [[ | ||
'''Consumer Information''' | '''Consumer Information''' | ||
| Line 75: | Line 90: | ||
[[Category:Profiles]] | [[Category:Profiles]] | ||
| + | [[Category:PaLM Profile]] | ||
| + | [[Category:DocShare]] | ||
| + | [[Category:CDA]] | ||
Latest revision as of 09:45, 14 April 2020
| This profile is part of the Pathology and Laboratory Medicine (PaLM) domain, which merged the former AP and LAB domains since 2016, January 4th. |
Sharing Laboratory Reports (XD-LAB) addresses the sharing of laboratory reports among a community of healthcare settings and care providers.
Summary
This Content Integration Profile describes a clinical laboratory report as an electronic document to be published towards a document sharing resource such as an Electronic Health Record (EHR) or a Personal Health Record (PHR) shared by a community of care providers, using one of the document sharing profiles defined in ITI-TF. Such an electronic document contains the set of releasable results produced by a clinical laboratory in fulfillment of one or more test Orders for a patient.
The report is both human-readable and importable in the consumer systems so as to consolidate their patient medical records.
The scope of this profile covers all laboratory specialties except anatomic pathology.
Benefits
Facilitates Patient Care Coordination
- Enables the caregiver to obtain at once the laboratory reports produced during previous episodes of care, which enhances continuity of care.
- Reduces over-ordering and over blood sampling from the patient by sharing the last laboratory reports among the various physicians attending the patient.
- Enables the physician to consolidate their patient biological history by importing into their EHR the structured and coded data embedded in the electronic laboratory report.
- Allows a public health laboratory to share broadly its reports related to a suspected outbreak
Details
The Sharing Laboratory Reports (XD-LAB) Integration Profile provides an electronic format for the laboratory report, to make this report:
- human-readable: displayed on screen or printed out, with a proper and clear layout and presentation.
- machine-readable: Each observation displayed in the report is also represented in the document as a set of structured and coded data that can be imported in the database of any consumer of the document.
This double capacity is achieved by leverageing the [Clinical Document Architecture Release 2] (CDAr2) standard from [HL7]
The XD-LAB Integration Profile covers six major use cases:
- At discharge time, a hospital physician publishing a summary laboratory report with the most significant results obtained during the patient stay.
- The bio-medical scientist of a private laboratory publishes a report for a patient into the regional Patient Health Record.
- An ambulatory physician shares a laboratory report obtained from a laboratory.
- A private or public laboratory publishes automatically all its reports in a shared document repository.
- A healthcare institution produces a cumulative report of all laboratory tests performed for the patient during the encounter.
- A public health laboratory shares its reports into a regional repository.
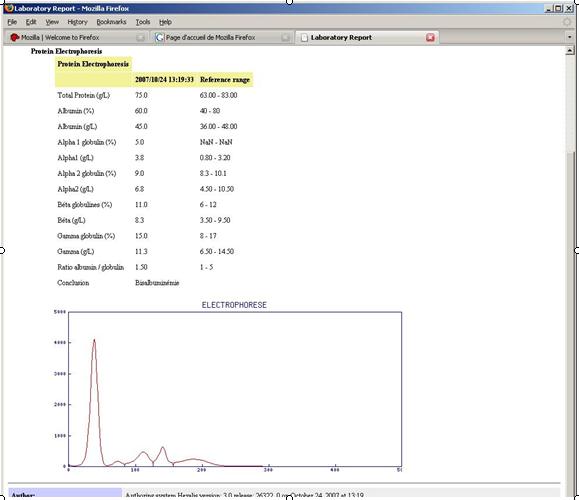
The standard CDAr2 leveraged by XD-LAB enables to share reports embedding images and charts, which are faithfully rendered by a web browser supporting RFC 2397:
Systems Affected
Systems involved in this profile are:
- A system owning a laboratory report and willing to publish it into a document sharing infrastructure. Depending upon the use cases, this can be the system of a clinical laboratory (LIS), the EHR of an ambulatory physician, the EMR of a care setting, the system of a public health laboratory. Each of these systems can play the role of Content Creator.
- The system operated by a professional caregiver who wants to view a laboratory report, and may want to import its structured and coded results into its own database. This system plays the role of Content Consumer.
Actors & Transactions:
Specification
Profile Status: Final Text
Documents: IHE Laboratory Technical Framework:
Computable specification:
- XD-LAB specified in Art-Decor on ihe-europe.net - templates and value sets
Underlying Standards:
See Also
Related Profiles
- Integration Profiles XD-LAB depends on
- Cross Enterprise Document Sharing (XDS) or Cross_Enterprise_Document_Media_Interchange (XDM) or Cross_Enterprise_Document_Reliable_Interchange (XDR) from the ITI Technical Framework. Each of these three profiles provides an infrastructure for the sharing of medical documents between the creators and the consumers.
- Audit Trail and Node Authentication (ATNA) from the ITI Technical Framework, to authenticate and audit the document-sharing transactions: Document storage and registration, document query and retrieval.
- Consistent Time (CT) from the ITI Technical Framework, to ensure accuracy of the time stamps in the shared documents and in the associated metadata.
Consumer Information
This page is based on the Profile Template