Comments
The Public Comment Phase of the development of each IHE Integration Profile allows interested people to provide input on the profiles, the scenarios and use cases they address, the technologies proposed and other details.
In particular, vendors who will implement the profiles and members of the healthcare community who will use the profiles are encouraged to review and comment.
- Review documents listed in the Public Comment columns on the Frameworks page.
- Note the deadline listed under the document link
- Submit your comments via the links provided at http://www.ihe.net/Public_Comment/.
The IHE Technical Committees who develop IHE Profiles will consider comments submitted in the specified public comment period and publish the disposition of and, where appropriate, a response to each comment.
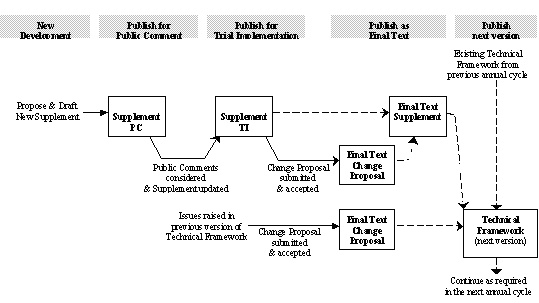
Phases of Development
Before any new Profile or other material is added to the IHE Technical Frameworks, it is written up as a separate "Supplement" document.
Public Comment - When new Supplements are sufficiently complete, they are published for 30 days of Public Comment. Comments are submitted via the links provided at http://www.ihe.net/Public_Comment/.
Trial Implementation - After the Public Comments have been reviewed, discussed and any necessary changes made, the Supplements are published for Trial Implementation.
Final Text - After Trial Implementation Supplements have been sufficiently implemented and tested and any necessary changes made, Supplements are published as Final Text. Final Text Supplements are merged into the domain Technical Framework document at the first opportunity, but are available until then as a separate document.
Retired - When a Supplement does not receive sufficient implementation, and is declared not mature it will be retired, also known as deprecated.
Change Proposals - After Supplements have been published (as Trial Implementation, as Final Text, or incorporated into a Technical Framework), changes are proposed and managed according to the CP Process.
The following diagram shows this process with some additional detail: