Laboratory Testing Workflow
| This profile is part of the Pathology and Laboratory Medicine (PaLM) domain, which merged the former AP and LAB domains since 2016, January 4th. |
Laboratory Testing Workflow (LTW) integrates the ordering, scheduling, processing, and result reporting activities associated with in vitro diagnostic tests performed by clinical laboratories in healthcare institutions.
Summary
The Laboratory Testing Workflow Profile covers the workflow related to tests performed on in vitro specimens by a clinical laboratory inside a healthcare institution, for both existing and pending orders, related to identified patients and unidentified or misidentified patients. It maintains the consistency of patient and order information from registration through ordering, scheduling, pre-analytical processing, testing, technical and clinical validation, to results reporting and usage of laoratory observations and comments by the care providers.
Benefits
Reduces Errors and Enhances Patient Care
- Prevents manual data entry errors by ensuring that each piece of information is entered only once, by the person closest to the source of this information, and from there, made available in due time to any system who needs it.
- Reduces over-ordering and over blood sampling from the patient by letting the current lab orders and their results follow the movements of the patient from a ward to another.
- Avoids lost specimens by tracking specimen collection in ward or in sample collection room, and specimen acceptance in laboratory.
- Accelerates the delivery of laboratory results to the ward staff, especially in emergency situations where a clinical decision is conditioned by some test results.
Improves Throughput in Care Process
- Suppresses manual order entry time on the laboratory side since the order is placed directly by the clinical system operated in the ward to the laboratory information system.
- Stremalines sample collection and transportation process, delivering a unique identifier to each specimen collected, tightly coupled with the identifiers of the orders requesting this specimen to be performed.
- Saves telephone calls between ward and laboratory since urgent results are delivered immediately and all status changes of any current order are reported in real time to the ordering system.
- Reduces the amount of paper reports since all lab results are consolidated in a global system that shares them with the authorized caregivers in the wards.
Favors independence of healthcare institution from their clinical and ancillary systems
- By providing clear specifications of interfaces between Clinical Information Systems (CIS), Laboratory Information Systems (LIS) and Laboratory Automation Systems (LAS), enables to replace one of these systems in a department or unit without breaking the existing data workflows involving this department or unit.
Details
The Laboratory Testing Workflow (LTW) Integration Profile is bound to in vitro diagnostic testing (tests performed on in vitro specimen collected from a patient). It supports all laboratory specialties but anatomic pathology.
Use cases:
The LTW Integration Profile covers three major use cases:
- Order placed with specimens collected and identified by the orderer
- Order placed with specimens collected by a third party, then identified and labeled by the laboratory information system
- Order generated by the laboratory and notified to the ordering system to obtain an order number.
Transactions:
LTW Integration Profile defines 5 Transactions, all of which based on HL7 release 2.5 or 2.5.1 messaging standard. Each transaction selects a subset of HL7 message structures (based on OML, ORL, ORU and OUL) and specifies a set of constraints applying to each message structure, as well as the responsibilities of the sender and receiver.
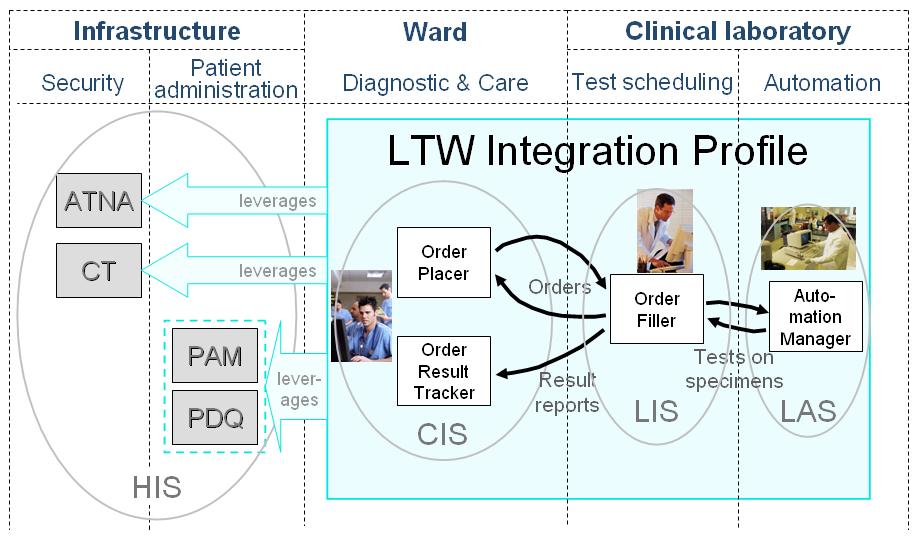
This profile leverages IT Infrastructure profiles [PAM] or [PDQ] for up to date patient and encounter data, and IT Infrastructure profiles [ATNA] and [CT] for security.
Units of work:
A laboratory request ordered by a physician for a patient is frequently composed of multiple tests and panels placed to a laboratory. Such a laboratory request containing several orders is called an Order Group in the HL7 standard, and uniquely identified by the ordering CIS with a Placer Group Number. Each of the orders is in turn uniquely identified by the CIS with a Placer Order Number. Each order accepted by the laboratory as well as each reflex order generated by the laboratory is uniquely identified by the LIS with a Filler Order Number.
Systems Affected
Systems involved in this profile are:
- One or more Clinical Information Systems (CIS). Each of these systems is operated by a clinical facility and provides a number of features such as order entry, order placing, placer order management and follow-up, order result tracking, management of patient biologic history, specimen calculation, specimen identification... A CIS usually implements the Actors Order Placer and Order Result Tracker.
- One or more Laboratory Information Systems (LIS). Each of these systems is operated by a number of clinical laboratories inside the institution. The LIS offers features such as order reception, specimen calculation, specimen identification or specimen acceptance, order check, scheduling, filler order management, production of worklists, result manual entry for non connected-tests, clinical validation and interpretation of results, result reporting. Each LIS implements an Order Filler Actor.
- One or more Laboratory Automation Systems (LAS) operated in each laboratory. A LAS manages a set of automated laboratory devices (pre-analytical devices, analyzers, post-analytical devices). The LAS receives Work Orders related to a specimen and processes the various steps of a Work Order on its set of devices, to eventually get the test results, perform the technical validation thereof, and upload them back to the LIS.
Actors & Transactions:
Specification
Profile Status: Final Text
Documents: IHE Laboratory Technical Framework:
Underlying Standards:
See Also
Related Profiles
- Integration Profiles LTW depends on
- Patient Administration Management (PAM) and/or Patient Demographics Query (PDQ) provide accurate patient demographics used by LTW: An application implementing one of the Actors Order Placer, Order Result Tracker, Order Filler shall also implement the Actors Patient Demographics Consumer and Patient Encounter Consumer from either PAM or PDQ Profile or both.
- Audit_Trail_and_Node_Authentication (ATNA) to audit creation and access to patient data during LTW. An application implementing one of the Actors Order Placer, Order Result Tracker, Order Filler shall also implement the Actor Secure Node from ATNA.
- Consistent_Time (CT) to ensure timestamps in LTW data and audit messages are accurate.
- Integration Profiles depending upon LTW
- Laboratory_Device_Automation (LDA) from the Laboratory Technical Framework: The Actor Automation Manager from the LDA Profile must also be an Automation Manager in the LTW Profile.
- Laboratory_Point_Of_Care_Testing (LPOCT) from the Laboratory Technical Framework: The Actor Order Filler from the Laboratory LPOCT Profile must also be an Order Filler in the LTW Profile.
- Laboratory_Barcode_Labeling (LBL) from the Laboratory Technical Framework: The Label Information Provider Actor of the LBL profile shall be grouped with either the Order Filler or the Order Placer from the LTW profile.
Consumer Information
This page is based on the Profile Template