Anatomic Pathology Structured Report
| This profile is part of the Pathology and Laboratory Medicine (PaLM) domain, which merged the former AP and LAB domains since 2016, January 4th. |
Summary
This APSR 2.0 IHE content profile specifies a unique digital document template for an anatomic pathology structured report (APSR) to be shared or exchanged between pathology laboratories and other care providers and institutions. This content profile describes how to exchange a digital anatomic pathology report in a human-readable format, which may include images. In addition it also contains findings and observations in a machine-readable structured format to facilitate the integration of individual observations and interpretations into the database of a consumer system, which will enable the application of automated reasoning on this content. The digital document template uses the HL7 Clinical Document Architecture standard (CDA R2).
Anatomic pathology structured reports document the findings on specimens removed from patients for diagnostic or therapeutic reasons covering all aspects of anatomic pathology (cancers, benign neoplasms as well as non-neoplastic conditions) as well as cytopathology. This information can be used for patient care, clinical research and epidemiology.
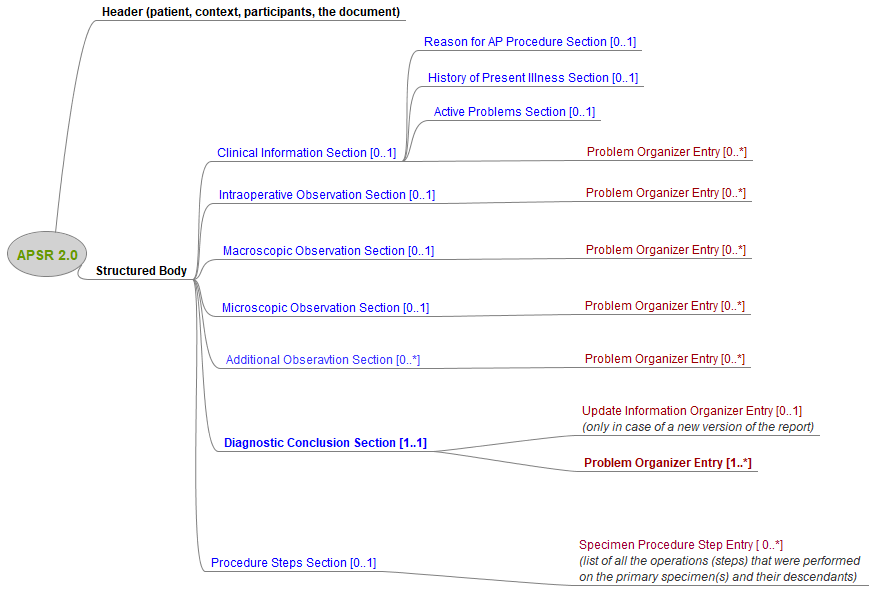
The document template includes a header conveying the contextual metadata of the report, and a structured body organized in sections. Each section contains a narrative content for the human reader and may also contain a number of entries (in red on the figure below) carrying the corresponding structured data intended to be integrated in the database of consuming applications.
Benefits
Standardizing and computerizing anatomic pathology reports is necessary to improve the quality of reporting and to facilitate the exchange and reuse of the content of these reports.
Details
The CDA document templated by APSR 2.0 is meant to be shared or exchanged by leveraging the document sharing profiles built by the IT Infrastructure domain of IHE.
The Clinical Information Section assembles the information delivered by the clinician who ordered to the pathology laboratory the diagnostic study that is documented in this report. The other sections report the activities, investigations and findings conducted by the laboratory. The only mandatory section in the report is the Diagnostic Conclusion Section, which conveys the conclusion of the pathology study both in human-readable format and in machine-processable format.
The optional Procedure Steps Section tracks all the activities of collection and preparation of specimens, and conveys the hierarchy of specimens from part to block, dice, ... to stained slide.
The structured data of most sections of the report is provided using the common template Problem Organizer Entry. An instance of this Entry assembles all the observations and related specimens, which investigate one problem identified in the study. e.g. "in situ carcinoma". Within the Organizer structure, the first observation describes the particular problem represented by this instance of Problem Organizer Entry.
The Update Information Organizer Entry is used only when a new revision of the report is shared, in replacement of a previous version. In this case this entry indicates the level of clinical significance of the update and lists the sections that have revised content.
Systems Affected
- Laboratory Information Systems (LIS) are usually the systems that produce anatomic pathology structured reports.
- HIS, CIS, EHR, EMR systems are the application that consume the reports.
- Other systems used by researchers or by public health institutions are potential consumers of these reports.
Specification
Profile Status: Trial Implementation
Documents: http://pubswiki.ihe.net/index.php/APSR20
Underlying Standards:
See Also
This page is based on the Profile Template