Lab AP Conf Minutes 2015-Sept-08: Difference between revisions
Jump to navigation
Jump to search
| Line 1: | Line 1: | ||
| | ||
<table> | <table> | ||
<tbody> | <tbody> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">Carolyn Knapik</td> | ||
<td width=" | <td width="128">CAP</td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">Francois Macary</td> | ||
<td width=" | <td width="128"> | ||
<table width="439"> | <table width="439"> | ||
<tbody> | <tbody> | ||
| Line 21: | Line 20: | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">Riki Merrick</td> | ||
<td width=" | <td width="128">APHL</td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">John Hopson</td> | ||
<td width=" | <td width="128">Abbott</td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">Naomi Ishii</td> | ||
<td width=" | <td width="128">JAHIS</td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">Ed Heierman</td> | ||
<td width=" | <td width="128">Abbott</td> | ||
</td> | |||
</tr> | </tr> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">Dmytro Rudd</td> | ||
<td width=" | <td width="128">Roche</td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">Benoit Denisselle</td> | ||
<td width=" | <td width="128"> </td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">Christel Daniel</td> | ||
<td width=" | <td width="128">AP-HP</td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">Joanna Selinsky</td> | ||
<td width=" | <td width="128">Beckman Coulter</td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">James Harrison</td> | ||
<td width=" | <td width="128">CAP</td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">Raj Dash, MD, FCAP</td> | ||
<td width=" | <td width="128">CAP</td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">Alessandro Sulis</td> | ||
<td width=" | <td width="128">CRS4</td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">Charles Parisot</td> | ||
<td width=" | <td width="128"> </td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
<td width=" | <td width="185">Laurent Lardin</td> | ||
<td width=" | <td width="128">BioMerieux</td> | ||
</tr> | </tr> | ||
</tbody> | </tbody> | ||
Revision as of 10:08, 9 September 2015
<tbody> </tbody>
| Carolyn Knapik | CAP | ||
| Francois Macary |
<tbody>
</tbody>
|
||
| Riki Merrick | APHL | ||
| John Hopson | Abbott | ||
| Naomi Ishii | JAHIS | ||
| Ed Heierman | Abbott | ||
| Dmytro Rudd | Roche | ||
| Benoit Denisselle | |||
| Christel Daniel | AP-HP | ||
| Joanna Selinsky | Beckman Coulter | ||
| James Harrison | CAP | ||
| Raj Dash, MD, FCAP | CAP | ||
| Alessandro Sulis | CRS4 | ||
| Charles Parisot | |||
| Laurent Lardin | BioMerieux |
Minutes
Joint activities
- Agenda Review:
- Timeslot for AP report progression:
- Still organizing getting together with the German colleagues – will aim for second hour of next month’s call or will send doodle– Note: October is the F2F visit in Tokyo
- German team has started to migrate the current revisions of APSR into Art Décor using a single AP observation template
- Ongoing effort to define top list of codes (for now PathLex codes to be migrated into LOINC) that can be used in CDA document – organ specific and general
- Next steps:
- Working on PSR document – have not managed to meet with the art décor team to create the new = this is a little ahead of current
- Take last release of APSR and migrate is as into Art Décor
- Update the next update of APSR based on the agreed CP for now
- The choice of the committee is to first migrate the current APSR specification into Art Décor, and then evaluate the output. Second, a distinct project (introduced through the regular “supplement proposal” process of IHE) will be to reuse this base material to build the “PSR” project, in the scope of the merged domain.
- Update on Merger Voting (Carolyn Knapik)
- Tally from last Thursday: 8 yes, 1 no – voting ends 9/16, so please vote, if you haven’t done so already
- Quorum for just IHE Lab was 4 – Carolyn will get quorum requirements from Chris Carr, might be hard to figure out which domain some folks belong to, as they belong to both, but get only 1 vote
- Update on LCC (James Harrison)
- Nothing new to report
- Managing distributed review and editing within Art Décor
- Easy to represent the entry templates, but unsure about how to build the larger value sets from SNOMED CT or LOINC – not specifically an issue for APSR, as the template has simple structure, but how to better represent the LOINC observation codes
- How far can we include SCT for value set generation – technical question:
- Building valuesets is possible in Art Décor – can be used to build from any reference terminology already loaded into the tool = ICD-10, LOINC, SCT (latest version of international)
- Would have to load PathLex into the tool, before we could use it to build out value sets based on PathLex
- Verify with German team
- Validate platform on clear scope of APSR first, then expand to PSR
- Prepare question(s) for Mary and Riki to present to board (Riki Merrick)
- Using Art décor as a publishing tool – what are the rules for changes and publication etc.
- How will Art Décor be connected to Gazelle tool for schematron – Eric has been working on a procedure to take the output of Art Décor and translate/ import into Gazelle tool (object checker – there is MOU between IHE Europe and Art Décor team)
- Where is the official instance of Art Décor for IHE?
- US is using an instance of Art Décor for a profile and Gazelle team is using that same instance
- Invite Eric to a next call to present the status of migration, possibly during the next face to face.
- Governance of Art Décor content?
- Feedback from DICOM Pathology meeting (Charles Parisot):
- Reviewed the proposed merger between AP and Lab – interested in tracking that – DICOM AP is supportive of this process and will monitor
- Plan for interoperability showcase(DICOM/IHE sponsored – working through the admin side of that) at May meeting of European College of Pathology in Berlin – workflow / including use of IHE profile – with possible repeat at CAP ‘16 in October – need more coordination
- Review of scenarios for the interoperability showcase – will have to create multiple, more complex scenarios that will use more IHE content in the long run
- F2F meeting logistics (Naomi Ishii)
- 9/19 – 21 – 3 full days – 9 AM– 5 PM local time
- Recommended hotels – Naomi to send on September 9
- Have meeting venue at JAHIS at Shinbashi station
- LAW (Ed Heierman)
- Status update:
- In progress on working with CLSI on creating the standard based on LAW – first draft due this month
- In process of collecting and creating final CPs prior to NA so that it can then be in TF (straightforward so far):
- Handling of profile option identifier
- Profile that should be optional on analyzer
- LAB-28 message structure choices
- OBX-4 usage
- Pooling of specimen
- Updatable / truncatable length
- Tracing inventory segments to one or more OBXs
- Minor edits to document
- LAW will be spotlighted for NA, also IICC will help with publicizing the NA connectathon t make this major testing event for LAW
- Gathering vendors for NA connectathon end of January 2016 in Cleveland
- For F2F hope to have all CPs reviewed and approved at end of the F2F in preparation for NA. We’ll allocate two time slots for this CP review during F2F.
- Technical Discussions
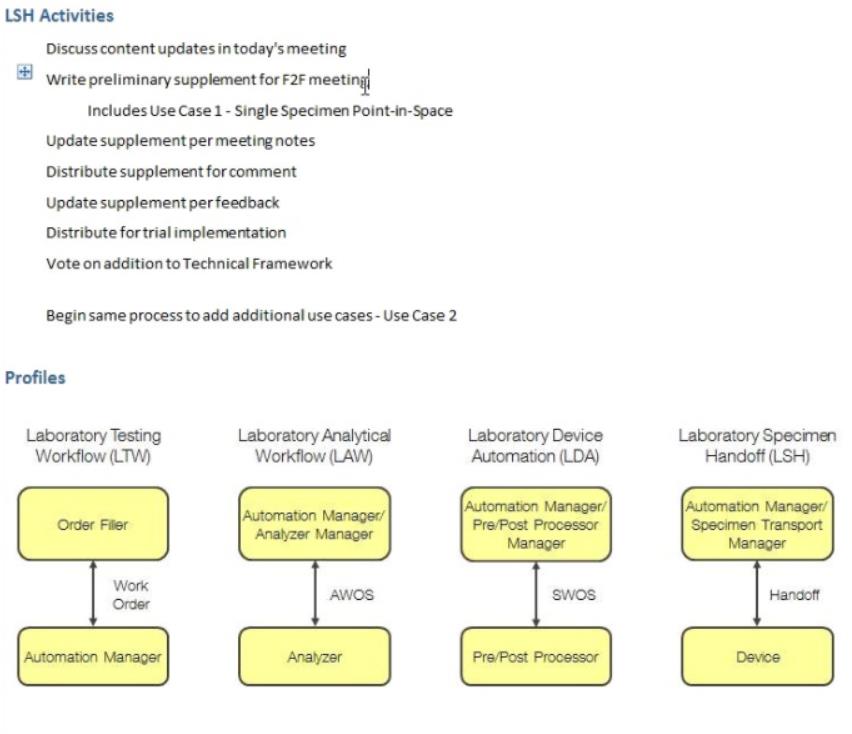
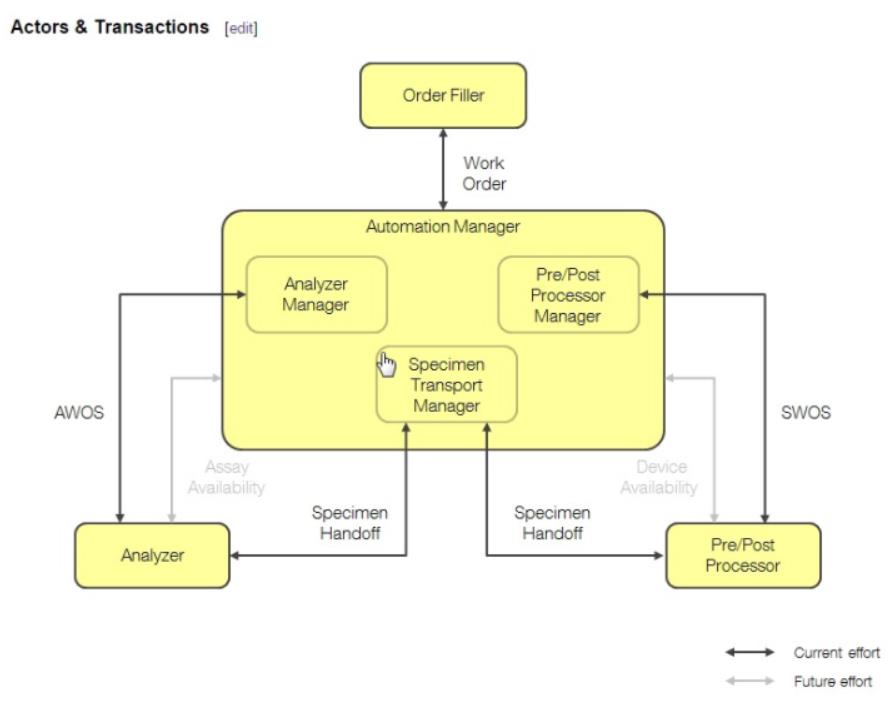
- LSH (John Hopson)
- Status
- LSH (John Hopson)
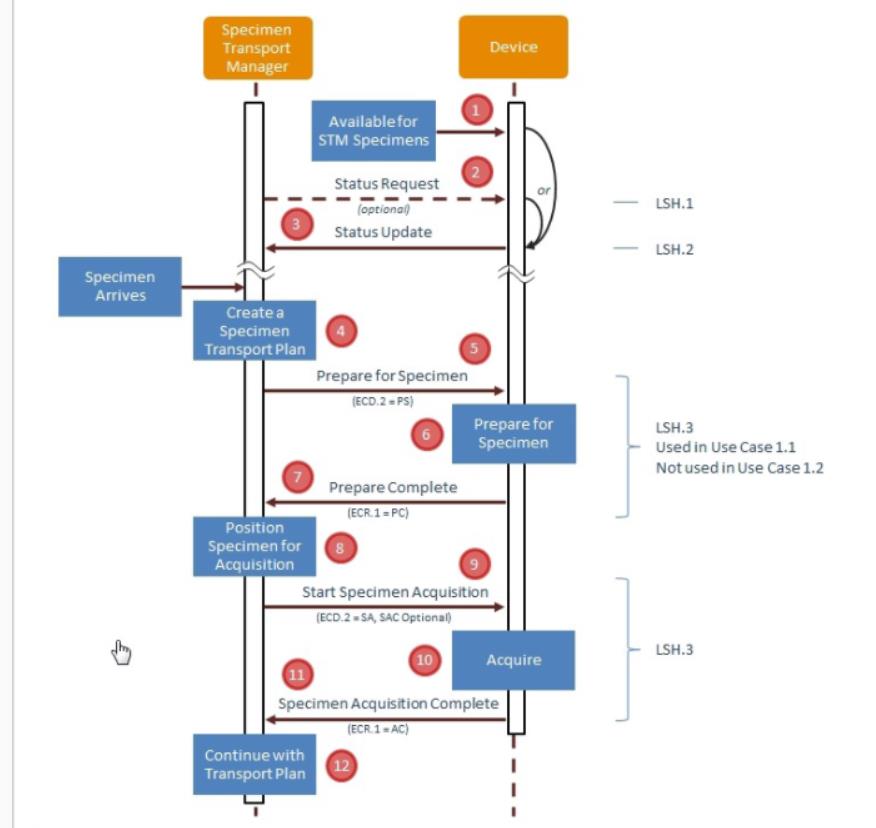
- So just 1 use case per Trial Implementation release - we have 1 year for testing and use case 1 MUST have enough interest of vendors to get proper testing – at least at NA, Europe and Japan connectathons
- Transactions
- Discuss HL7 Carrier/Tray Status Messages
- Message Structure
- Have 3 specific transactions
- Reviewing command and response:
- SAC segment where device identifies the specimen can be omitted – so changed usage in LSH.3 to RE
- Reviewing Chapter 13 – how do you communicate carrier and tray status to the automation manager – have specimen status and container status SAC-8 - can identify the tray / container, but nothing specifically to the status of the carrier / tray – ask Andrzej Knafel, main author of Chapter 13
- Type of statuses: complete processing, etc – Dmytro will draft suggestion for these new fields – John will provide the detail Dmytro would need to write up a CR for OO
- OO usually meets on Thursday 1 PM ET – Riki will let him know when it is on the agenda
- F2F meeting needs: 3-4 hours – may a 3 hour block plus 1 hour next day for follow up work?
- Who will attend October F2F in person?
- Dmytro via webex
- Alessandro in person
- Riki via webex only most likely
- Will send out draft agenda and an RSVP request to the list
Call adjourned 10:50 AM EDT