Laboratory Barcode Labeling: Difference between revisions
| (17 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
'''Laboratory Specimen Barcode Labeling(LBL)''' | {| style="color: blue;" | ||
| '''This profile is part of the Pathology and Laboratory Medicine (PaLM) domain, which merged the former AP and LAB domains since 2016, January 4th.''' | |||
|} | |||
'''Laboratory Specimen Barcode Labeling (LBL)''' supports the robotization of specimen container identification and delivery at blood sample collection time, in the context of laboratory test requests. | |||
__TOC__ | __TOC__ | ||
| Line 6: | Line 9: | ||
==Summary== | ==Summary== | ||
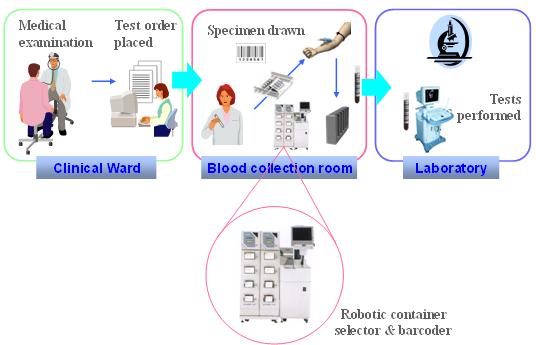
The Laboratory Specimen Barcode Labeling(LBL) Integration Profile supports this workflow: A robotic system delivers specimen containers pre-identified with a bar coded label, for the specimen collection related to a laboratory test request. This robotic system receives patient, test order and specimen data from another system (HIS, CIS or LIS depending upon the organization), and issues a label for each specimen container needed, with the specimen identifier bar coded on the label, and possibly other information printed out on this label. | The Laboratory Specimen Barcode Labeling (LBL) Integration Profile supports this workflow: A robotic system delivers specimen containers pre-identified with a bar coded label, for the specimen collection related to a laboratory test request. This robotic system receives patient, test order and specimen data from another system (HIS, CIS or LIS depending upon the organization), and issues a label for each specimen container needed, with the specimen identifier bar coded on the label, and possibly other information printed out on this label. | ||
<br/> | <br/> | ||
The robotic system is operated in the blood sample collection room. Some healthcare institutions have a central specimen collection room shared by a number of clinical wards. Other institution decentralize the blood sampling process in each clinical ward. This LBL profile supports both kinds of organizations. | The robotic system is operated in the blood sample collection room. Some healthcare institutions have a central specimen collection room shared by a number of clinical wards. Other institution decentralize the blood sampling process in each clinical ward. This LBL profile supports both kinds of organizations. | ||
<br/> | |||
<center>[[Image:Ihe_lab_lbl_ip.jpg]]</center> | |||
<br/> | <br/> | ||
| Line 16: | Line 21: | ||
* Identifies the specimen container at blood sample collection time, with an identifier tightly coupled to the laboratory request this specimen is for, and to the patient subject of the request. | * Identifies the specimen container at blood sample collection time, with an identifier tightly coupled to the laboratory request this specimen is for, and to the patient subject of the request. | ||
* Robotizes and accelerates blood sample collection process. | * Robotizes and accelerates blood sample collection process. | ||
* Reduces patient waiting time in the blood sample collection room. | |||
* Avoids barcode reading problems on downstream analyzers. | |||
==Details== | ==Details== | ||
The '''''Laboratory | The '''''Laboratory Specimen Barcode Labeling (LBL) Integration Profile''''' covers '''5 use cases''' reflecting five possible organizations: | ||
* | * Specimen container identification and delivery process driven by the clinical ordering system (HIS/CIS/EMR), in push mode. | ||
* | * Specimen container identification and delivery process driven by the laboratory information system (LIS), in push mode. | ||
* | * Specimen container identification and delivery process driven by HIS/CIS/EMR, from specimen data provided by LIS, in push mode. | ||
* Specimen container identification and delivery process driven by HIS/CIS/EMR, in pull mode. | |||
* | * Specimen container identification and delivery process driven by LIS, in pull mode. | ||
* | The LBL Integration Profile defines '''2 Transactions''', all of which based on HL7 release 2.5 or 2.5.1 messaging standard: | ||
* LAB-61 Label Delivery Request (push mode) | |||
The | * LAB-62 Query for Label Delivery Instructions (pull mode) | ||
* LAB- | <br/> | ||
* LAB- | |||
==Systems Affected== | ==Systems Affected== | ||
Systems involved in this profile are: | Systems involved in this profile are: | ||
* The LIS or HIS or CIS/EMR, which will request the specimen provision, depending upon the enterprise organization. This system will be the Label Information Provider Actor. | |||
* The specimen containers provider, which is a robotic system managing containers, a barcode printer and stickers automatically stuck to containers. This system will be the Label broker Actor. | |||
'''Actors & Transactions:''' | '''Actors & Transactions:''' | ||
<center>[[Image: | <center>[[Image:lbl-actor-transaction.JPG]]</center> | ||
<br/> | <br/> | ||
==Specification== | ==Specification== | ||
'''Profile Status:''' [[Comments| | '''Profile Status:''' [[Comments| Final Text]] | ||
'''Documents:''' | '''Documents:''' [http://www.ihe.net/Technical_Framework/index.cfm#laboratory IHE Laboratory Technical Framework:] | ||
:* [http://www.ihe.net/Technical_Framework/upload/ihe_lab_tf_rel2.pdf Vol. 1] - Section 7 (LBL profile overview) | |||
:* [http://www.ihe.net/Technical_Framework/upload/ihe_lab_tf_rel2-2.pdf Vol. 2] - Sections 16 and 17 (LBL transactions descriptions) | |||
'''Underlying Standards:''' | '''Underlying Standards:''' | ||
:* [http://www.hl7.org HL7] | :* [http://www.hl7.org HL7] | ||
| Line 64: | Line 63: | ||
'''Related Profiles''' | '''Related Profiles''' | ||
* '''Integration Profiles | * '''Integration Profiles LBL depends on''' | ||
:* [[Laboratory Testing Workflow]] (LTW) | :* [[Laboratory Testing Workflow]] (LTW) which provides the order details necessary for correct labeling. The Actor Label Information Provider in LBL profile shall be grouped with one of the Actors Order Placer or Order Filler in the LTW profile. | ||
| Line 73: | Line 72: | ||
[[Category:Profiles]] | [[Category:Profiles]] | ||
[[Category:PaLM Profile]] | |||
[[Category:HL7v2]] | |||
Latest revision as of 05:28, 1 August 2017
| This profile is part of the Pathology and Laboratory Medicine (PaLM) domain, which merged the former AP and LAB domains since 2016, January 4th. |
Laboratory Specimen Barcode Labeling (LBL) supports the robotization of specimen container identification and delivery at blood sample collection time, in the context of laboratory test requests.
Summary
The Laboratory Specimen Barcode Labeling (LBL) Integration Profile supports this workflow: A robotic system delivers specimen containers pre-identified with a bar coded label, for the specimen collection related to a laboratory test request. This robotic system receives patient, test order and specimen data from another system (HIS, CIS or LIS depending upon the organization), and issues a label for each specimen container needed, with the specimen identifier bar coded on the label, and possibly other information printed out on this label.
The robotic system is operated in the blood sample collection room. Some healthcare institutions have a central specimen collection room shared by a number of clinical wards. Other institution decentralize the blood sampling process in each clinical ward. This LBL profile supports both kinds of organizations.
Benefits
Improves integrity and efficiency of in vitro diagnostic testing in healthcare institutions
- Identifies the specimen container at blood sample collection time, with an identifier tightly coupled to the laboratory request this specimen is for, and to the patient subject of the request.
- Robotizes and accelerates blood sample collection process.
- Reduces patient waiting time in the blood sample collection room.
- Avoids barcode reading problems on downstream analyzers.
Details
The Laboratory Specimen Barcode Labeling (LBL) Integration Profile covers 5 use cases reflecting five possible organizations:
- Specimen container identification and delivery process driven by the clinical ordering system (HIS/CIS/EMR), in push mode.
- Specimen container identification and delivery process driven by the laboratory information system (LIS), in push mode.
- Specimen container identification and delivery process driven by HIS/CIS/EMR, from specimen data provided by LIS, in push mode.
- Specimen container identification and delivery process driven by HIS/CIS/EMR, in pull mode.
- Specimen container identification and delivery process driven by LIS, in pull mode.
The LBL Integration Profile defines 2 Transactions, all of which based on HL7 release 2.5 or 2.5.1 messaging standard:
- LAB-61 Label Delivery Request (push mode)
- LAB-62 Query for Label Delivery Instructions (pull mode)
Systems Affected
Systems involved in this profile are:
- The LIS or HIS or CIS/EMR, which will request the specimen provision, depending upon the enterprise organization. This system will be the Label Information Provider Actor.
- The specimen containers provider, which is a robotic system managing containers, a barcode printer and stickers automatically stuck to containers. This system will be the Label broker Actor.
Actors & Transactions:
Specification
Profile Status: Final Text
Documents: IHE Laboratory Technical Framework:
Underlying Standards:
See Also
Related Profiles
- Integration Profiles LBL depends on
- Laboratory Testing Workflow (LTW) which provides the order details necessary for correct labeling. The Actor Label Information Provider in LBL profile shall be grouped with one of the Actors Order Placer or Order Filler in the LTW profile.
Consumer Information
This page is based on the Profile Template