Laboratory Device Automation: Difference between revisions
New page: '''Laboratory Device Automation(LDA)''' Integration Profile supports the workflow on the automated technical section of the clinical laboratory. The automated section of a laboratory can b... |
→Specification: updated link from LAB TF to PaLM TF |
||
| (21 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
'''Laboratory Device Automation(LDA)''' | {| style="color: blue;" | ||
| '''This profile is part of the Pathology and Laboratory Medicine (PaLM) domain, which merged the former AP and LAB domains since 2016, January 4th.''' | |||
|} | |||
'''Laboratory Device Automation (LDA)''' supports workflow in the automated technical section of the clinical laboratory, also known as the "work area". | |||
__TOC__ | __TOC__ | ||
| Line 6: | Line 9: | ||
==Summary== | ==Summary== | ||
The Laboratory Device Automation (LDA) Integration Profile | The Laboratory Device Automation (LDA) Integration Profile covers the exchanges between an Automation Manager (Actor played by a Laboratory Information System '''LIS''' or by a Laboratory Automation System '''LAS''') and a set of automated Laboratory Devices (LD) to process a Work Order, perform the tests on the related specimens and retrieve their results. This processing includes the pre-analytical process of the specimen (sorting, centrifugation, aliquoting, transportation, decapping) the analytical process itself (run of the ordered tests on the specimen) and the post-analytical process (recapping, transportation, rerun, storage and retrieval). | ||
<br/> | <br/> | ||
The scope of this LDA profile is strictly restricted to automated devices operated inside a clinical laboratory. Whenever the same devices are used on the point of care and operated by the ward staff, they are taken care of by the | The scope of this LDA profile is strictly restricted to automated devices operated inside a clinical laboratory. Whenever the same devices are used on the point of care and operated by the ward staff, they are taken care of by the [[Laboratory Point Of Care Testing]] Profile. | ||
<br/> | <br/> | ||
The input of the workflow is a Work Order: A set of tests requested on one or more specimens, to be fulfilled by the Automation Manager. This Actor splits the Work Order into one or more Work Order Steps (WOS) | The input of the workflow is a '''Work Order''': A set of tests requested on one or more specimens, to be fulfilled by the Automation Manager. This Actor splits the Work Order into a sequence of one or more '''Work Order Steps (WOS)'''. | ||
<br/>Each WOS represents one action to be performed by one device on one specimen. | |||
<br/> | <br/> | ||
<center>[[Image:Ihe_lab_lda_ip.jpg]]</center> | <center>[[Image:Ihe_lab_lda_ip.jpg]]</center> | ||
| Line 17: | Line 21: | ||
==Benefits== | ==Benefits== | ||
''' | '''Improves efficiency of the clinical laboratory''' | ||
* | * Streamlines specimen processing in the work area of the clinical laboratory. | ||
* Reduces the processing time and accelerates the delivery of test results. | |||
'''Favors independence of laboratories from their automated devices''' | |||
* By providing clear specifications of interfaces between Laboratory Automation System (LAS), and the various types of automated devices this profile enables a laboratory to replace a device by a new one without breaking the existing workflow in the work area. | |||
* | |||
'''Favors independence of | |||
* By providing clear specifications of interfaces between | |||
==Details== | ==Details== | ||
The '''''Laboratory | The '''''Laboratory Device Automation (LDA) Integration Profile''''' covers six major use cases: | ||
* WOS downloaded on the device before specimen arrival | |||
* Query for the WOS at specimen arrival on the device | |||
* | * Manual entry of the WOS on the LD | ||
* | * Rerun on the Analyzer, automatically launched by the analyzer or requested by the LAS at technical validation time. | ||
* | * Quality Control order downloaded by the LAS | ||
* Quality Control order queried by the device | |||
* Unsolicited QC results sent by the device. | |||
The LDA Integration Profile defines 4 Transactions, all of which based on HL7 release 2.5 or 2.5.1 messaging standard: | |||
* LAB-21 WOS download to automated device | |||
* LAB-22 WOS queried by automated device | |||
* LAB-23 Status change of an analytical WOS, and upload of the results produced | |||
* LAB-26: Stautus change of a pre-analytical or post-analytical WOS | |||
Each of these transaction selects a subset of HL7 message structures (based on OML, ORL and OUL) and specifies a set of constraints applying to each message structure, as well as the responsibilities of the sender and receiver.<br/> | |||
==Systems Affected== | ==Systems Affected== | ||
| Line 44: | Line 48: | ||
Systems involved in this profile are: | Systems involved in this profile are: | ||
* | * The Actor Automation Manager can be played either by a Laboratory Automation System ('''LAS'''), which is a system dedicated to the work area of the laboratory, or by the Laboratory Information System ('''LIS'''), which usually supports the capacity to connect some if not all the automated devices of the work area. In frequent organizations a laboratory may combine a LIS and one or more LAS, each system handling a subset of devices. | ||
* This profile distinguishes two categories of automated devices: | |||
* | :* The pre/pos-analytical devices (centrifuge, robotic specimen conveyer, decapper, robotized specimen storage, aliquoter... | ||
:* The analyzer, which performs tests and produces observations related to a specimen. | |||
<br/> | |||
'''Actors & Transactions:''' | '''Actors & Transactions:''' | ||
<center>[[Image: | <center>[[Image:lda-actor-transaction.JPG]]</center> | ||
<br/> | <br/> | ||
==Specification== | ==Specification== | ||
'''Profile Status:''' [[Comments| | '''Profile Status:''' [[Comments| Final Text]] | ||
'''Documents:''' [https://www.ihe.net/resources/technical_frameworks/#PaLM IHE Pathology and Laboratory Medicine Technical Framework:] | |||
'''Underlying Standards:''' | '''Underlying Standards:''' | ||
:* [http://www.hl7.org HL7] | :* [http://www.hl7.org HL7] | ||
| Line 70: | Line 72: | ||
'''Related Profiles''' | '''Related Profiles''' | ||
* '''Integration Profiles | * '''Integration Profiles LDA depends on''' | ||
:* [[ | :* [[Laboratory Testing Workflow]] (LTW): The Actor Automation Manager shall also be an Automation Manager in the LTW profile. Thus, it will fulfill Work Order, received in the LTW profile, handling each Work Order as a sequence of Work Order Steps performed in the LDA profile | ||
'''Consumer Information''' | '''Consumer Information''' | ||
| Line 83: | Line 81: | ||
[[Category:Profiles]] | [[Category:Profiles]] | ||
[[Category:PaLM Profile]] | |||
[[Category:HL7v2]] | |||
Latest revision as of 14:34, 17 November 2022
| This profile is part of the Pathology and Laboratory Medicine (PaLM) domain, which merged the former AP and LAB domains since 2016, January 4th. |
Laboratory Device Automation (LDA) supports workflow in the automated technical section of the clinical laboratory, also known as the "work area".
Summary
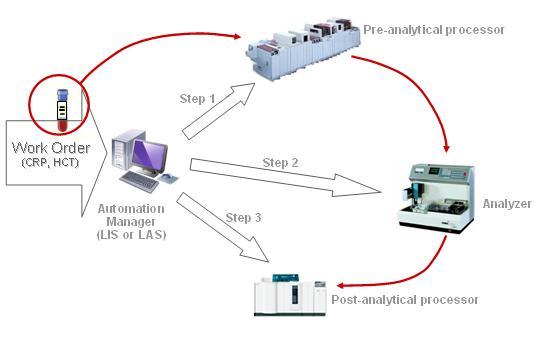
The Laboratory Device Automation (LDA) Integration Profile covers the exchanges between an Automation Manager (Actor played by a Laboratory Information System LIS or by a Laboratory Automation System LAS) and a set of automated Laboratory Devices (LD) to process a Work Order, perform the tests on the related specimens and retrieve their results. This processing includes the pre-analytical process of the specimen (sorting, centrifugation, aliquoting, transportation, decapping) the analytical process itself (run of the ordered tests on the specimen) and the post-analytical process (recapping, transportation, rerun, storage and retrieval).
The scope of this LDA profile is strictly restricted to automated devices operated inside a clinical laboratory. Whenever the same devices are used on the point of care and operated by the ward staff, they are taken care of by the Laboratory Point Of Care Testing Profile.
The input of the workflow is a Work Order: A set of tests requested on one or more specimens, to be fulfilled by the Automation Manager. This Actor splits the Work Order into a sequence of one or more Work Order Steps (WOS).
Each WOS represents one action to be performed by one device on one specimen.
Benefits
Improves efficiency of the clinical laboratory
- Streamlines specimen processing in the work area of the clinical laboratory.
- Reduces the processing time and accelerates the delivery of test results.
Favors independence of laboratories from their automated devices
- By providing clear specifications of interfaces between Laboratory Automation System (LAS), and the various types of automated devices this profile enables a laboratory to replace a device by a new one without breaking the existing workflow in the work area.
Details
The Laboratory Device Automation (LDA) Integration Profile covers six major use cases:
- WOS downloaded on the device before specimen arrival
- Query for the WOS at specimen arrival on the device
- Manual entry of the WOS on the LD
- Rerun on the Analyzer, automatically launched by the analyzer or requested by the LAS at technical validation time.
- Quality Control order downloaded by the LAS
- Quality Control order queried by the device
- Unsolicited QC results sent by the device.
The LDA Integration Profile defines 4 Transactions, all of which based on HL7 release 2.5 or 2.5.1 messaging standard:
- LAB-21 WOS download to automated device
- LAB-22 WOS queried by automated device
- LAB-23 Status change of an analytical WOS, and upload of the results produced
- LAB-26: Stautus change of a pre-analytical or post-analytical WOS
Each of these transaction selects a subset of HL7 message structures (based on OML, ORL and OUL) and specifies a set of constraints applying to each message structure, as well as the responsibilities of the sender and receiver.
Systems Affected
Systems involved in this profile are:
- The Actor Automation Manager can be played either by a Laboratory Automation System (LAS), which is a system dedicated to the work area of the laboratory, or by the Laboratory Information System (LIS), which usually supports the capacity to connect some if not all the automated devices of the work area. In frequent organizations a laboratory may combine a LIS and one or more LAS, each system handling a subset of devices.
- This profile distinguishes two categories of automated devices:
- The pre/pos-analytical devices (centrifuge, robotic specimen conveyer, decapper, robotized specimen storage, aliquoter...
- The analyzer, which performs tests and produces observations related to a specimen.
Actors & Transactions:
Specification
Profile Status: Final Text
Documents: IHE Pathology and Laboratory Medicine Technical Framework:
Underlying Standards:
See Also
Related Profiles
- Integration Profiles LDA depends on
- Laboratory Testing Workflow (LTW): The Actor Automation Manager shall also be an Automation Manager in the LTW profile. Thus, it will fulfill Work Order, received in the LTW profile, handling each Work Order as a sequence of Work Order Steps performed in the LDA profile
Consumer Information
This page is based on the Profile Template