|
|
| (8 intermediate revisions by one other user not shown) |
| Line 1: |
Line 1: |
| The ADX (Aggregate Data Exchange) profile affords data exchange partners a way to define a Data Structure Definition and the associated normative schema of the data message for a particular indicator report. To foster further adoption, the Aggregate Data Exchange-HIV (ADX-HIV) profile leverages the ADX profile to develop a content specification specifically for HIV.
| | |
|
| |
|
| __TOC__ | | __TOC__ |
|
| |
|
| ==Summary== | | ==Summary== |
| ''<ADX-HIV Profile leverages on the ADX Profile version 2.1, which is currently published for trial implementation. It aims to set down a concrete example of how to leverage ADX profile for interoperability in health information systems used for managing and reporting on HIV.> ''
| | The ADX (Aggregate Data Exchange) profile affords data exchange partners a way to define a Data Structure Definition and the associated normative schema of the data message for a particular indicator report. To foster further adoption, the Aggregate Data Exchange-HIV (ADX-HIV) profile leverages the ADX profile version 2.1, which is currently published for trial implementation, to develop a content specification specifically for HIV. |
| | |
| ''<Insert a simple graphic that, at a glance, visually summarizes what the profile is about. Do not use an actor/transaction diagram here. Show your graphic to someone for 5 seconds (literally) and ask them what it's about. If what they say hits the main points in your summary paragraph, you have succeeded. E.g. a graphic of a hospital, a clinic, and a lab with patient records moving between them. .>''
| |
|
| |
|
| ''<See [[Help:Contents#Tips_.26_Tricks| Help - Tips and Tricks]] for details on inserting an image/graphic.>''
| | ADX-HIV is a Content Profile that defines a common Data Structure Definition (DSD) with structural metadata (codelists with data elements and associated disaggregation) to represent data elements HIV core indicators that could be readily used in existing systems for reporting. It aims to set down a concrete example of how to leverage ADX profile for interoperability in health information systems used for managing and reporting on HIV towards achieving epidemic control and the 90-90-90 global goals. These goals, to be accomplished by 2020, are: for 90% of people with HIV to be diagnosed and know their status; for 90% of people living with HIV (PLHIV) to be on Antiretroviral Therapy (ART); and for 90% of people on ART to be virally suppressed. |
|
| |
|
| ==Benefits== | | ==Benefits== |
| ''<If the profile can improve Cost, Safety, Quality or Efficiency then list the specific examples of that benefit (e.g. error reduction, increased throughput) and how they come about (e.g. SWF reduces patient errors due to mistyped demographics at the modality by transfering demographics electronically from the Order Filler). Consider using a bullet list for readability. Such benefits help users and vendors make the business case for the profile. If the profile does not improve any aspect of Cost, Safety, Quality or Efficiency feel free to talk about something else here.>''
| | ADX-HIV will help govern and enforce interoperability between HIV point of care (source) systems and HMIS (receiver) systems. This will reduce the burden within jurisdictions for developing HIV core indicators schema. It will also foster harmonization of HIV core indicators across jurisdictions and increase the ability to compare results between health facilities, subnational levels and even countries. |
|
| |
|
| ==Details== | | ==Details== |
|
| |
|
| ''<A few paragraphs, if appropriate, providing more details (mostly in user-speak, not tech-speak) on what the profile does and how it works.>''
| | This ADX-HIV profile defines a common DSD for HIV core indicators with data elements and associated disaggregations along with instructions regarding how to leverage these to generate and exchange conformant XML messages. Interoperable message exchange will be valuable to jurisdictions such as Ministries of Health, and WHO, and to health initiatives such as the U.S. President’s Emergency Plan for AIDS Relief (PEPFAR), the Global Fund, UNAIDS, and others (see figure below). |
|
| |
|
| ''<If the user might be familiar with the mechanisms used by the profile, you can mention them here. E.g. Evidence Documents is based on DICOM Structured Report (SR) Templates.>''
| | [[File:ADX-HIV_image.png]] |
|
| |
|
| ''<If the user might have an appreciation for the problems addressed in the profile, you can mention them here, but keep it short. E.g. Mapping HL7 Order fields to DICOM Modality Worklist attributes can be inconsistent in the marketplace, so Scheduled Workflow provides vendors with more detailed instructions.>''
| |
|
| |
| ==Systems Affected==
| |
| ''<List (in user terms) the types of systems they might expect to have implemented actors from this profile, e.g. RIS, PACS, HIS, CAD Workstation, etc. and for each, how it would participate.>''
| |
|
| |
|
| * ''PACS systems may store, manage, and/or display Evidence Documents.''
| | A jurisdiction can extend the ADX-HIV DSD and validation schemas to meet additional HIV reporting requirements specific to their context. |
| * ''Display systems may query, retrieve and display Evidence Documents.''
| |
| * ''Reporting workstations may retrieve, process and include details from Evidence Documents in reports
| |
|
| |
|
| '''Actors & Transactions:'''
| | ==Systems Affected== |
| | | * <b>National Health Management Information Systems</b> would make use of ADX to receive HIV routine reports from health facilities and other national systems. |
| ''<Insert an actor-transaction diagram, and or list of Content Definitions>''
| | * <b>EMR</b> systems at health facilities might use ADX to create HIV monthly reports. |
| | * International organizations might use ADX to harmonize HIV reporting between regions and countries. |
|
| |
|
| ==Specification== | | ==Specification== |
|
| |
|
| '''Profile Status:''' [[Comments| Final Text]] | | '''Profile Status:''' Public Comments |
| ''<Replace "Final Text" with "Trial Implementation" or "Public Comment" as appropriate.>''
| |
|
| |
|
| '''Documents:''' | | '''Documents:''' |
|
| |
| ''<Provide direct links to the specific volumes or supplements, and list the volume sections relevant to this profile. This is a simple inventory of official normative and informative text. If you would like to provide a reading guide or walkthrough of what is in each of the different sections for implementers or users, do that in the Profile FAQ or the Profile Implementation Page linked below. If the profile uses transactions from multiple Tech. Frameworks, repeat the structure below.>''
| |
|
| |
| [http://www.ihe.net/Technical_Framework/index.cfm#radiology IHE Radiology Technical Framework:]
| |
| :* [http://www.ihe.net/Technical_Framework/upload/ihe_tf_rev8.pdf Vol. 1] - Section 5 (SWF Profile)
| |
| :* [http://www.ihe.net/Technical_Framework/upload/ihe_tf_rev8-2.pdf Vol. 2] - Sections 4.8 to 4.10, 4.14 to 4.19, and 4.23
| |
| :* [http://www.ihe.net/Technical_Framework/upload/ihe_tf_rev8-3.pdf Vol. 3] - Appendix E
| |
|
| |
|
| '''Underlying Standards:''' | | '''Underlying Standards:''' |
| | * ADX Profile [https://ihe.net/uploadedFiles/Documents/QRPH/IHE_QRPH_Suppl_ADX.pdf ADX Supplement] |
| | * ISO 8601 |
| | * SNOMED-CT |
| | * HL7 |
|
| |
|
| ''<list all the standards on which the profile is based; if possible with links to sources>''
| |
| :* [http://dicom.nema.org DICOM]
| |
| :* [http://www.hl7.org HL7]
| |
| :* ...
| |
|
| |
| ==See Also==
| |
|
| |
| ''<The following sections can be left out if there is nothing to point to. This is just to show where such information can go.>''
| |
|
| |
|
| |
| '''Related Profiles'''
| |
|
| |
| ''<List profiles this one depends on, profiles that depend on this one, profiles that are synergistic with this one. Start with the name of the other profile as a link and then explain the relationship.>''
| |
|
| |
| * ''[[Reporting Workflow]] [RWF] may use Evidence Documents as inputs to the reporting process.''
| |
| * ''[[Simple Image & Numeric Reports]] [SINR] may include data copied from Evidence Documents.''
| |
| * ''[[Cross-enterprise Document Sharing for Imaging]] [XDS-I] can be used to share Evidence Documents between sites over a network.''
| |
| * ''[[Portable Data for Imaging]] [PDI] can store Evidence Documents on media such as CDs.''
| |
| * ''[[Import Reconciliation Workflow]] [IRWF] can fix patient ids, etc. of Evidence Documents when importing.''
| |
|
| |
|
| |
| '''Consumer Information'''
| |
|
| |
| The [[Profile FAQ Template]] answers typical questions about what the Profile does. ''<Replace the link with a link to the actual FAQ page for the Profile>''
| |
|
| |
| The [[Profile Purchasing Template]] describes considerations when purchasing equipment to deploy this Profile. ''<Replace the link with a link to the actual Purchasing page for the Profile>''
| |
|
| |
| '''Implementer Information'''
| |
|
| |
| The [[Profile Implementation Template]] provides additional information about implementing this Profile in software. ''<Replace the link with a link to the actual Implementation page for the Profile>''
| |
|
| |
| '''Reference Articles'''
| |
|
| |
| ''<List References (good and bad) (with link if possible) to Journal Articles that mention IHE's work (and hopefully include some analysis). Go ahead, Google: IHE <Profile Name> abstract or Google: IHE <Profile Name> and under the "more" select "Scholar". You might be surprised. >''
| |
|
| |
|
| [[Category:Profiles]] | | [[Category:Profiles]] |
|
| |
| This page is based on the [[Profile Overview Template]]
| |
|
| |
|
| |
| <!--
| |
| Categorize the Domain of your Profile with one of the following (delete the rest):
| |
| [[Category:CARD Profile]]
| |
| [[Category:DENT Profile]]
| |
| [[Category:ENDO Profile]]
| |
| [[Category:EYECARE Profile]]
| |
| [[Category:ITI Profile]]
| |
| [[Category:PaLM Profile]]
| |
| [[Category:PCC Profile]]
| |
| [[Category:PCD Profile]]
| |
| [[Category:PHARM Profile]]
| |
| [[Category:QRPH Profile]] | | [[Category:QRPH Profile]] |
| [[Category:RO Profile]] | | [[Category:ADX]] |
| [[Category:RAD Profile]]
| |
| | |
| Categorize key standards used in your Profile with one or more of the following (delete the rest):
| |
| [[Category:CDA]]
| |
| [[Category:FHIR]]
| |
| [[Category:DICOM]]
| |
| [[Category:DICOMweb]]
| |
| [[Category:HL7v2]]
| |
| [[Category:XDW]]
| |
| [[Category:DocShare]]
| |
| | |
| And if people forget to do this, we'll comment this out and you can forget to opt-in. :-)
| |
| -->
| |
Summary
The ADX (Aggregate Data Exchange) profile affords data exchange partners a way to define a Data Structure Definition and the associated normative schema of the data message for a particular indicator report. To foster further adoption, the Aggregate Data Exchange-HIV (ADX-HIV) profile leverages the ADX profile version 2.1, which is currently published for trial implementation, to develop a content specification specifically for HIV.
ADX-HIV is a Content Profile that defines a common Data Structure Definition (DSD) with structural metadata (codelists with data elements and associated disaggregation) to represent data elements HIV core indicators that could be readily used in existing systems for reporting. It aims to set down a concrete example of how to leverage ADX profile for interoperability in health information systems used for managing and reporting on HIV towards achieving epidemic control and the 90-90-90 global goals. These goals, to be accomplished by 2020, are: for 90% of people with HIV to be diagnosed and know their status; for 90% of people living with HIV (PLHIV) to be on Antiretroviral Therapy (ART); and for 90% of people on ART to be virally suppressed.
Benefits
ADX-HIV will help govern and enforce interoperability between HIV point of care (source) systems and HMIS (receiver) systems. This will reduce the burden within jurisdictions for developing HIV core indicators schema. It will also foster harmonization of HIV core indicators across jurisdictions and increase the ability to compare results between health facilities, subnational levels and even countries.
Details
This ADX-HIV profile defines a common DSD for HIV core indicators with data elements and associated disaggregations along with instructions regarding how to leverage these to generate and exchange conformant XML messages. Interoperable message exchange will be valuable to jurisdictions such as Ministries of Health, and WHO, and to health initiatives such as the U.S. President’s Emergency Plan for AIDS Relief (PEPFAR), the Global Fund, UNAIDS, and others (see figure below).
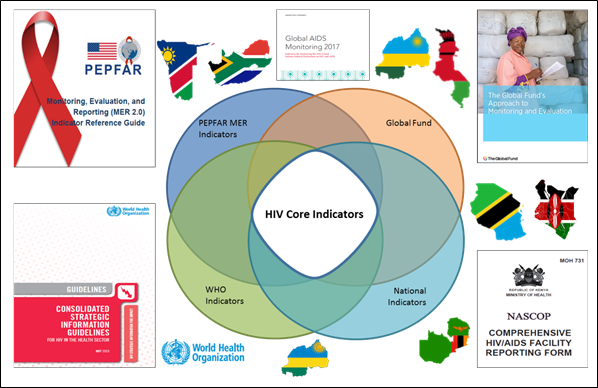
A jurisdiction can extend the ADX-HIV DSD and validation schemas to meet additional HIV reporting requirements specific to their context.
Systems Affected
- National Health Management Information Systems would make use of ADX to receive HIV routine reports from health facilities and other national systems.
- EMR systems at health facilities might use ADX to create HIV monthly reports.
- International organizations might use ADX to harmonize HIV reporting between regions and countries.
Specification
Profile Status: Public Comments
Documents:
Underlying Standards:
