Point-of-Care Medical Device Tracking (PMDT): Difference between revisions
Tsoutherland (talk | contribs) m Tsoutherland moved page Point-of-Care Medical Device Tracking to Point-of-Care Medical Device Tracking (PMDT): add |
|||
| (57 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
==Summary== | |||
=='''Summary''' == | |||
The Point-of-Care Medical Device Tracking (PMDT) Profile closes the loop on data acquisition at the point-of-care in support of reporting data about implantable medical devices (e.g., pacemaker, titanium plates) and medical devices (e.g., vital sign monitors, pulse oximeters, blood glucose monitors) during a procedure (e.g., Continuous Pulse Oximetry - 4A19XCZ, Insertion Pacemaker - 0JH607Z, Open Reduction Internal Fixation Elbow - 0PSJ04Z). The approach to acquiring the medical device information at the point-of-care enables it to be retrieved and reused at a later time. | The Point-of-Care Medical Device Tracking (PMDT) Profile closes the loop on data acquisition at the point-of-care in support of reporting data about implantable medical devices (e.g., pacemaker, titanium plates) and medical devices (e.g., vital sign monitors, pulse oximeters, blood glucose monitors) during a procedure (e.g., Continuous Pulse Oximetry - 4A19XCZ, Insertion Pacemaker - 0JH607Z, Open Reduction Internal Fixation Elbow - 0PSJ04Z). The approach to acquiring the medical device information at the point-of-care enables it to be retrieved and reused at a later time. | ||
[[Image:PMDT_General_IHE_wiki_Image.png|250px]] | |||
==Benefits== | |||
The benefits of this profile are: | ''' | ||
== '''Benefits''' == | |||
'''The benefits of this profile are: | |||
• '''Closes the loop on data acquisition at the point-of-care to support reporting of medical device data''' | • '''Closes the loop on data acquisition at the point-of-care to support reporting of medical device data''' | ||
:o Medical device data can be exchanged with: | |||
:: * Other providers for continuum of care (e.g., Discharge Summary, Referrals) | |||
:: * Registries (e.g., Total Joint Registry) | |||
:: * Payers for reimbursement (e.g., government provided, private insurance) | |||
o | |||
:o Associating a medical device used for monitoring a disease or symptom of a disease (e.g., vital sign monitors, pulse oximeters, blood glucose monitors) to a patient for: | |||
:: * Querying the device or procedure using the medical devices UDI | |||
:: * Accurately matching the device data with the correct patient | |||
'' | • '''Increase patient safety''' | ||
:o Traceability of medical devices (avoid use of counterfeit or illegitimate products) | |||
:o Quality issues identified (e.g., recalls, adverse events) | |||
• '''Increase accurate medical device data capture at the point-of-care''' | |||
:o Eliminates human error from manual medical device data entry by using the devices unique device identifier (UDI) supplied in a structured format from the manufacturer | |||
:o Provides a standards-based mechanism to capture medical device information consistently across the care setting'' | |||
== '''Details''' == | |||
Implantable medical devices are essential for the treatment and management of a wide variety of medical conditions. These devices are costly and concerns about illegitimate (i.e., counterfeit, stolen) products being used for patient care has become a global issue. Post-market surveillance of implantable medical devices can be challenging due to the longevity of the patient and the medical device unless there is a reliable implant tracking method. Using the HL7 FHIR StructureDefinition Resources (HL7 FHIR Device, HL7 FHIR Procedure Resource) to record information about medical devices (the unique device identifier [UDI]), including implantable/life-supporting/life-sustaining devices and tissues, provides a reliable and standards-based mechanism to perform a search and query function by using the HL7 FHIR RESTful Services (HTTP/HTTPS) to create/update and query existing records. | |||
'' | =='''Systems Affected'''== | ||
* Enterprise EHRs | |||
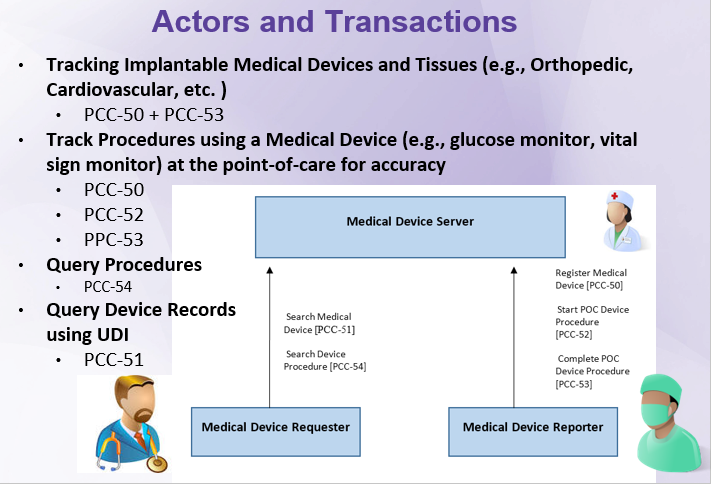
=='''Actors & Transactions:''' == | |||
[[Image:PMDTwikipageActorsTransactions_Image.png|400px]] | |||
=='''Specification''' == | |||
: | '''Profile Status:''' [[Comments| Trial Implementation]] | ||
* https://www.ihe.net/uploadedFiles/Documents/PCC/IHE_PCC_Suppl_PMDT.pdf | |||
'''Documents:''' | |||
[http://www.ihe.net/Technical_Framework/index.cfm# Patient Care Coordination (PCC) IHE PCC Technical Framework:] | |||
:* [http://ihe.net/uploadedFiles/Documents/PCC/IHE_PCC_TF_Vol1.pdf Vol. 1] - Integration Profiles | |||
:* [http://ihe.net/uploadedFiles/Documents/PCC/IHE_PCC_TF_Vol2.pdf Vol. 2] - Transactions and Content Modules | |||
'''Underlying Standards:''' | |||
* | :* FHIR STU3 [http://www.hl7.org/fhir] | ||
=='''See Also'''== | |||
IHE PCD-01 transactions based on HL7 v2.7 ORU_R01 | |||
'''Related Profiles''' | |||
* IHE PCC Remote Patient Monitoring (RPM) Profile | |||
[[Category:Profiles]][[Category:Patient Care Coordination]] | |||
[[Category:FHIR]] | [[Category:FHIR]] | ||
Latest revision as of 18:20, 11 December 2020
Summary
The Point-of-Care Medical Device Tracking (PMDT) Profile closes the loop on data acquisition at the point-of-care in support of reporting data about implantable medical devices (e.g., pacemaker, titanium plates) and medical devices (e.g., vital sign monitors, pulse oximeters, blood glucose monitors) during a procedure (e.g., Continuous Pulse Oximetry - 4A19XCZ, Insertion Pacemaker - 0JH607Z, Open Reduction Internal Fixation Elbow - 0PSJ04Z). The approach to acquiring the medical device information at the point-of-care enables it to be retrieved and reused at a later time.
Benefits
The benefits of this profile are:
• Closes the loop on data acquisition at the point-of-care to support reporting of medical device data
- o Medical device data can be exchanged with:
- * Other providers for continuum of care (e.g., Discharge Summary, Referrals)
- * Registries (e.g., Total Joint Registry)
- * Payers for reimbursement (e.g., government provided, private insurance)
- o Associating a medical device used for monitoring a disease or symptom of a disease (e.g., vital sign monitors, pulse oximeters, blood glucose monitors) to a patient for:
- * Querying the device or procedure using the medical devices UDI
- * Accurately matching the device data with the correct patient
• Increase patient safety
- o Traceability of medical devices (avoid use of counterfeit or illegitimate products)
- o Quality issues identified (e.g., recalls, adverse events)
• Increase accurate medical device data capture at the point-of-care
- o Eliminates human error from manual medical device data entry by using the devices unique device identifier (UDI) supplied in a structured format from the manufacturer
- o Provides a standards-based mechanism to capture medical device information consistently across the care setting
Details
Implantable medical devices are essential for the treatment and management of a wide variety of medical conditions. These devices are costly and concerns about illegitimate (i.e., counterfeit, stolen) products being used for patient care has become a global issue. Post-market surveillance of implantable medical devices can be challenging due to the longevity of the patient and the medical device unless there is a reliable implant tracking method. Using the HL7 FHIR StructureDefinition Resources (HL7 FHIR Device, HL7 FHIR Procedure Resource) to record information about medical devices (the unique device identifier [UDI]), including implantable/life-supporting/life-sustaining devices and tissues, provides a reliable and standards-based mechanism to perform a search and query function by using the HL7 FHIR RESTful Services (HTTP/HTTPS) to create/update and query existing records.
Systems Affected
- Enterprise EHRs
Actors & Transactions:
Specification
Profile Status: Trial Implementation
Documents:
Patient Care Coordination (PCC) IHE PCC Technical Framework:
Underlying Standards:
- FHIR STU3 [1]
See Also
IHE PCD-01 transactions based on HL7 v2.7 ORU_R01
Related Profiles
- IHE PCC Remote Patient Monitoring (RPM) Profile