Quality Measure Execution-Early Hearing
The Quality Measure Execution-Early Hearing (QME-EH) profile describes the content needed to communicate patient-level data to electronically monitor the performance of early hearing detection and intervention (EHDI) initiatives for newborns.
Part of the Early Hearing Detection and Intervention - Family of Profiles
Summary
The Newborn Hearing Screening measure is a process measure conducted as a part of the U.S. Centers for Disease Control and Prevention (CDC) Early Hearing Detection and Intervention (EHDI) public health program. It measures the proportion of newborns who receive hearing screening prior to discharge at birth.
The Quality Measure Execution-Early Hearing (QME-EH) Content Profile outlines steps to create and consume standard electronic patient-level and aggregate-level quality reports for the Newborn Hearing Screening (CMS31v4) electronic clinical quality measure (eCQM). QME-EH provides methods for reusing data from a standard summary of care document, generated by an EHR to create a patient-level quality report. Additionally, it specifies how to create an aggregate-level quality report for the Newborn Hearing Screening quality measure using multiple patient-level quality reports.
This profile specifies information exchange methods which permit greater data transparency and consistency for the quality measurement process and which reduce the burden of compliance with quality measurement programs.
This profile does not specify how to determine if the set of documents (clinical summary documents or patient-level quality reports) supplied for processing is the correct and complete set of documents to be processed for the measure. Actors creating quality reports need to determine if a document that is supplied in the run meets the measure definition’s criteria for the initial population of the measure before processing the rest of the data. Data in documents which meet the initial population (IP) criteria should be included in the quality report. Refer to QRPH TF-3: X.6.3 for considerations regarding the use of a mechanisms defined within the IHE QRPH Newborn Admission Notification Information (NANI) Profile to confirm if the run of documents processed for the quality measure is complete.
Benefits
- Ensures digital newborn hearing screening quality measure data are captured and communicated, supporting consistent quality measure computation
- Standardizes quality measurement for newborn care in birthing facilities
- Provides interoperability between clinical EHRs and Public Health EHDI Information Systems (EHDI-IS) for increased efficiency and data quality
- Reduces reporting burden for EHRs by allowing quality measure data to be consumed out of standard clinical summaries
Details
The Quality Measure Execution-Early Hearing (QME-EH) content profile specifies how to use the HL7 Quality Reporting Document Architecture (QRDA) standard to implement a patient-level quality measure report for newborn hearing screening.
QRDA is a specialized implementation guide (IG) for the HL7 Clinical Document Architecture standard (CDA R2). It establishes general guidelines for creating three types of CDA documents for sharing different types of quality measure reports.
The QME-EH profile shows implementers how to use a supplied measure definition for newborn hearing screening, and the measure definition’s associated data set and data processing logic, to create and consume a CDA-based patient-level or aggregate-level quality measure report.
Systems Affected
Figure X.1-1 shows the actors directly involved in the QME-EH Profile and the direction that the content is exchanged. The technical actor initiating information sharing has options for creating a Summary of Care Document, a Patient-Level Quality Report, or an Aggregate-Level Quality Report. The technical actor consuming the information similarly has the same options. This flexible design permits the burden of reporting to be shared across different systems with varying levels of quality measure computation functionality.
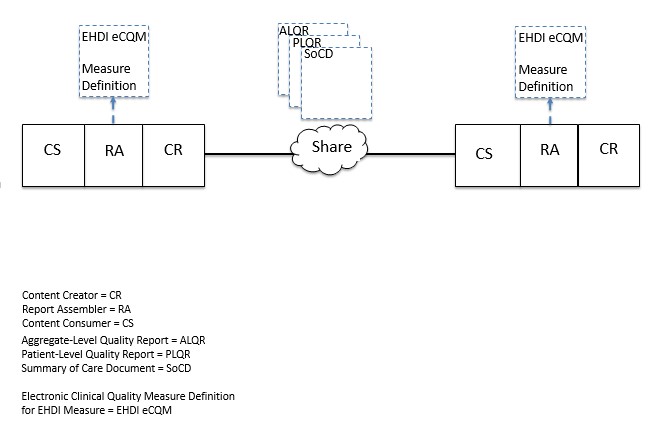
Note: The Actor Diagram for this profile is modular in nature. Options are used to indicate the grouping requirements/capabilities of a system participating in the information exchange supporting creation of the quality measure reports. The actor options for a participating system are determined by the role the system intends to play in a particular use case for this profile (see Section X.2 and X.3). Use Cases in Section X.3 include customized diagrams which specify the actor options needed to support the various use cases. Section X.4.1 contains additional information about the concepts behind the modular grouping options in this profile.
A product implementation using this profile must group actors from this profile with actors from a workflow or transport profile to be functional. The grouping of the content module described in this profile to specific actors is described in more detail in the “Required Actor Groupings” section of the IHE QRPH Supplement QME-EH.
Table X.1-1 lists the content module(s) defined in the QME-EH Profile. To claim support with this profile, an actor shall support all required content modules (labeled “R”) and may support optional content modules (labeled “O”).
| Actors | Content Modules (See Note 1) | Optionality | Reference |
|---|---|---|---|
| Report Assembler | EHDI Measure Definition (eCQM EDHI) | R | QRPH TF-1:Appendix C |
| Content Consumer (See Note 1) | Summary of Care Document (SoCD) | O | A C-CDA Clinical summary. For the US Realm this is a CCD or Discharge Summary which includes information needed to populate the data elements defined for a PLQR. (See QRPH TF-4: Appendix D.1.1) |
| Content Consumer (See Note 1) | Aggregate-Level Quality Report (ALQR) | O | For US Realm, see QRPH TF-4:R1.3.1.1.D2 |
| Content Consumer (See Note 1) | Patient-Level Quality Report | O | For US Realm, see QRPH TF-4:R1.3.1.1.D1 |
| Content Creator (See Note 1) | Summary of Care Document (SoCD) | O | Any C-CDA clinical summary such as a CCD or Discharge Summary which includes information needed to populate the data elements defined for a PLQR. For US Realm, see QRPH TF-4:Appendix D.1.1 |
| Content Creator (See Note 1) | Aggregate-Level Quality Report (ALQR) | O | For US Realm, see QRPH TF-4:R1.3.1.1.D2 |
| Content Creator (See Note 1) | Patient-Level Quality Report (PLQR) | O | For US Realm, see QRPH TF-4:R1.3.1.1.D1 |
Options that may be selected for each actor in this profile, if any, are listed in Table X.2-1. Dependencies between options when applicable are specified in notes.
| Actor | Option Name | Reference |
|---|---|---|
| Content Creator (See Note 1) | Aggregate-Level Quality Report (ALQR) Option | QRPH TF-1:X.2.1 |
| Content Creator (See Note 1) | Patient-Level Quality Report (PLQR) Option | QRPH TF-1:X.2.2 |
| Content Creator (See Note 1) | Summary of Care Document (SoCD) Option | QRPH TF-1:X.2.3 |
| Content Consumer (See Note 2) | Aggregate-Level Quality Report (ALQR) Option | QRPH TF-1:X.2.1 |
| Content Consumer (See Note 2) | Patient-Level Quality Report (PLQR) Option | QRPH TF-1:X.2.2 |
| Content Consumer (See Note 2) | Summary of Care Document (SoCD) Option (See Note 3) | QRPH TF-1:X.2.3 |
| Report Assembler (See Note 4) | Assemble Patient-Level Quality Report from Summary of Care Document (SoCD) to PLQR) Option | QRPH TF-1:X.2.4 |
| Report Assembler (See Note 4) | Assemble Aggregate-Level Quality Report from Patient-Level Quality Report (PLQR to ALQR) Option | QRPH TF-1:X.2.5 |
Specification
Profile Status: Trial Implementation
Underlying Standards:
- HL7 Clinical Document Architecture (CDA® Release 2)
- HL7 QRDA R3 Jun 2105
- HL7 QDM-Based HQMF R1 Feb 2014
- Logical Observation Identifiers Names and Codes (LOINC®)
- Systematized Nomenclature Of Medicine Clinical Terms (SNOMED-CT)
This page is based on the Profile Overview Template