QRPH 12Mar08 Minutes
The Domain recapped discussions from 11 March to review options regarding profile(s) for 2008.
Domain Profile Status Discussion:
- The measure specification standard is not clear. QRDA is not scheduled to incorporate the requirement in the expected September DSTU, Care Plans as an option may not be sufficient. The Collaborative effort to define the measure specification model will not be complete until approximately October 2008 and it is not incorporated within an standard to date.
- Measure output has risks if QRDA is limited to the existing 3 measures and if it does not successfully achieve DSTU status as of September.
- We have option of profiling the American Heart Association ‘Get with the Guidelines’ or American College of Cardiology National Cardiovascular Data Registry based on work done in the HL7 Cardiology SIG.
- There is also an option using the XDS sharing value sets. A content profile to address the data and terminology for public health is an option for a profile. There is currently a project occurring in 3 countries in the Middle East. In discussion it was determined that further evaluation be performed to creating content profiles for value sets to enable use by query definers once query / measure definition infrastructure can be determined as a profile. Content profiles have benefit for the short term in that any representation of a query or quality measure requires reference to population identifiers as oids, data element requirements as oids and exclusion criteria as oids. By having query (measure) developers use such oids in their specifications has near term benefit for creation of queries directly within clinical records.
- Value set consumer - 2008
- Value set repository - 2008
- Needed Value set registry - Re-evaluate for 2009
- We need to learn more about a potential request regarding an aggregate reporting output in collaboration with work being done in NY State. Further details pending.
- The Medication adverse event reporting profile will be presented Thursday AM.
- Content profile to incorporate required reporting data set on newborns and children. The example is a requirement from France in that hospitals must report at 8 days, 9 months and 24 months a specific data set to Public Health. There are similar requirements in Australia. The US as a dataset reporting requirement called EPSDT (Early Periodic Screening and Diagnostic Testing) for Medicaid children. This is a potential profile that could be constrained based in individual country requirements.
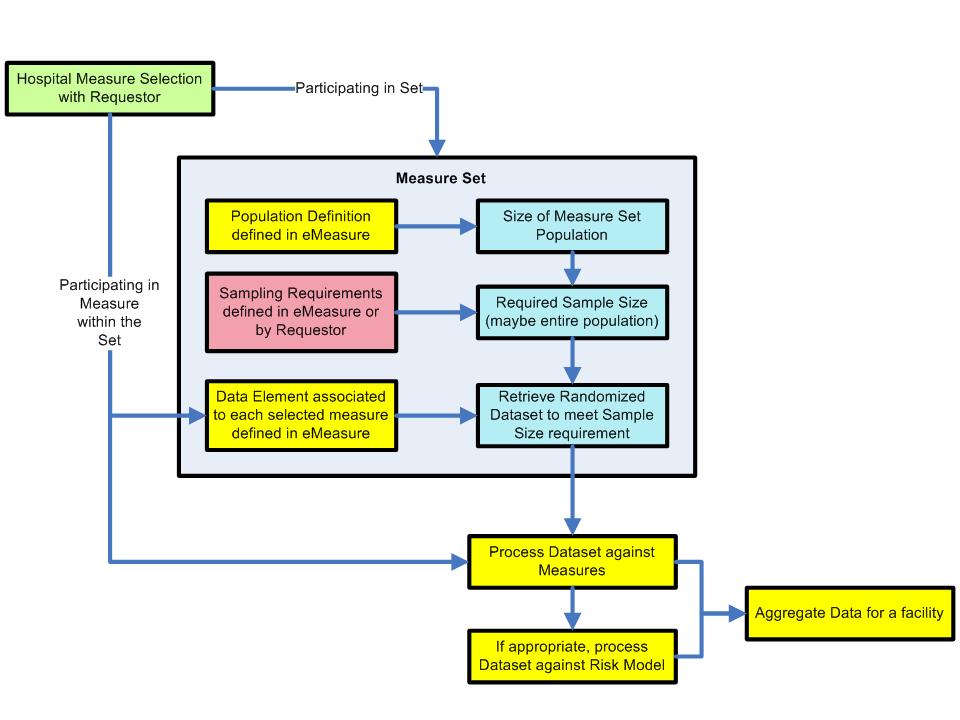
The specification of a clinical measure was identified as a high priority option. In that regard, the workflow requirement issues were discussed with input from Patty Craig, Associate Project Director, Center for Data Management of the Joint Commission.
The Joint Commission sends a measure selection list to the vendors every night. Not clear if changes are identified from one version to the next. i.e., what version and what is different from the prior version. Some vendors have automated the selection screen by which hospitals select the measures with which they will participate. Issue is identification of which measures are selected by the site. By selecting measures the provider opts in to participate.
Each organization (e.g., hospital) has the option of negotiating with Joint Commission to determine if it will participate in each set, or each measure within a set (participation in set statement in above diagram). Definitions of measure sets (etc.) as per the US Agency for Healthcare Research and Quality (AHRQ) is provided below. It is not clear if Joint Commission will maintain sets or revert to individual measures, or sets that contain multiple populations. For IHE, we need to consider [1] :
- Ability to provide a specification
- A Collection is the highest possible level of the measure hierarchy. A Collection may contain one or more Sets, Subsets, Composites, and/or individual Measures.
- A Set is the next level of the hierarchy. A Set may include one or more Subsets, Composites, and/or individual Measures.
- A Subset is the third level of the hierarchy. A Subset may include one or more Composites, and/or individual Measures.
- A Composite is a measure whose score is an aggregate of scores from other measures. A Composite may include one or more other Composites and/or individual Measures. Composites may be comprised of Measures that can or cannot be used on their own; this information is provided in the "DESCRIPTION" field of the Measure Summaries.
- A Measure is the lowest level of the hierarchy and may belong to a Composite, Subset, Set, and/or Collection.
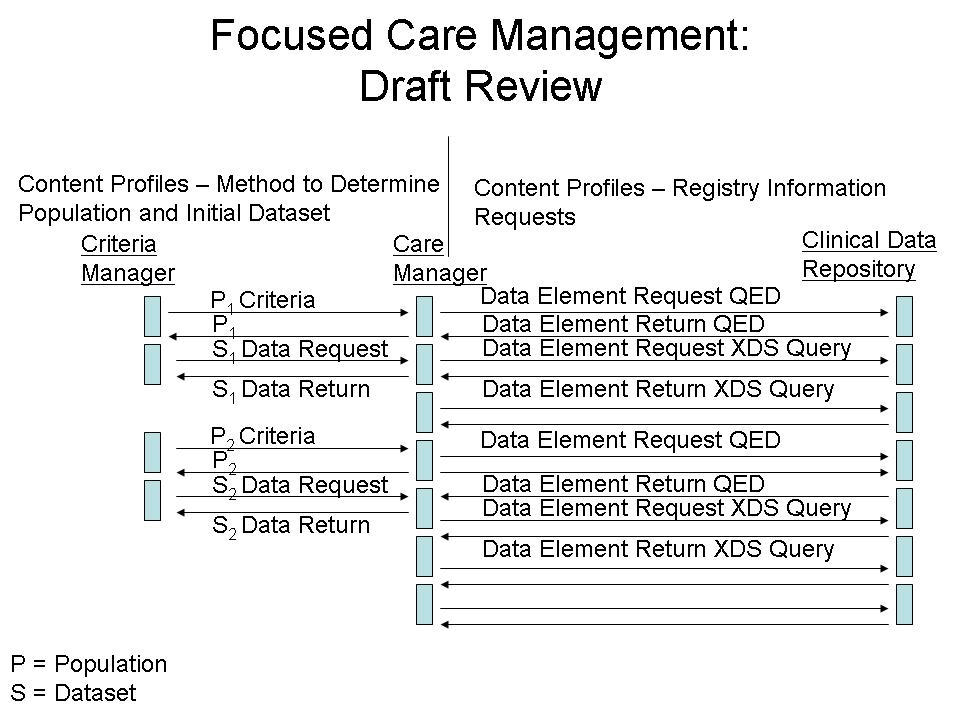
The "Population Definition" identified in the figure above, is defined within the measure workflow (Focused Care Management Draft) shown in the figure below (P1).
Some other concepts to be considered with respect to the measure workflow:
- Time
- Time frame of data collection – as queries / measures are updated on a semi-annual basis, the population definition may vary (i.e., P1 may become P2 for definition of the population part way through a measurement cycle). Similarly the data requirement may change during the measurement cycle.
- Time frame for selection of population
- Inpatient hospital measures – based on discharge or potentially events that occur during a cycle
- Outpatient hospital measures – based on the encounter
- Ambulatory measures – based on occurrence of a procedure during the cycle, or potentially based on membership of the individual in the practice and/or health plan during the entire period of measurement. The may be represented by definition of medical home, number of visits during the measurement period, etc.
- Sample size definition
- Is the provider sampling or sending all data?
- Sampling can be performed by set, subset or measure level in that the sample is based on the applicable population for that set, subset or measure and may be different for others.
- Sampling requirements must be part of the metadata of the measure
- Sampling could be defined for the measure, subset or set and all requestors may not use the same sampling method.
Plans for 13 March 2008 - Review Drug Safety Content Profile, Select and create action plan around profiles for 2008.