PaLM Conf Minutes 2022-January-12
Jump to navigation
Jump to search
Attendees
| Name | |
|---|---|
| Alessandro Sulis | alessandro.sulis@crs4.it |
| Jim McNulty | jmcnult@cap.org |
| Mary Kennedy | mkenned@cap.org |
| Dan Rutz | drutz@epic.com |
| Raj Dash | r.dash@duke.edu |
| David DeMena | |
| Jim Harrison | james.harrison@virginia.edu |
| Francesca Frexia | francesca.frexia@crs4.it |
| JD Nolen | jdlnolen@gmail.com |
| Gianluca Pavan | Arsenal.IT |
| Gunter Haroske | haroske@icloud.com |
| Filip Migom | filip.migom@gmail.com |
| Nick Haarselhorst | nick.haarselhorst@philips.com |
| Kevin Schap | kschap@cap.org |
| Megumi Kondo | megumi.kondo.sakura.japan@gmail.com |
| Jurgen Brandstatter | j.brandstaetter@codewerk.at |
| Hynek Kruzik | kruzik@gnomon.cz |
| Francois Macary | Francois.macary@phast.fr |
| Riki Merrick | rikimerrick@gmail.com |
Next meeting: February 9, 2022
- Administration:
- F2F: The group will stick with the dates currently scheduled, 5/2-5/4, 2022
- Wiki changes needed:
- Milestones for 2021 and 2022 need to do this before the IHE PaLM Board report is finalized this week.
- Dates are needed for the publication matrix for 2022.
- As a group, we should make sure EVERY published profile has a wiki page using the correct template
- IHE PaLM Board report:
- Alessandro will check on participation numbers for Connectathon participation in 2021.
- The tables will be combined into the first table.
- Riki will send a draft to the email list - please provide feedback by the end of this week.
- DP-AT project update
- The white paper will be sent out by email to provide feedback. Please send feedback by the next PaLM committee call
- Please share the document as a pdf and send comments back to Gianluca
- The challenge of vendors receiving DP-AT because of PACs system drive workflow was discussed.
- HL7 Germany
- DEMIS FHIR specs are the official spec for reporting micro to PH starting tomorrow.
- APSR work is separate from DEMIS FHIR
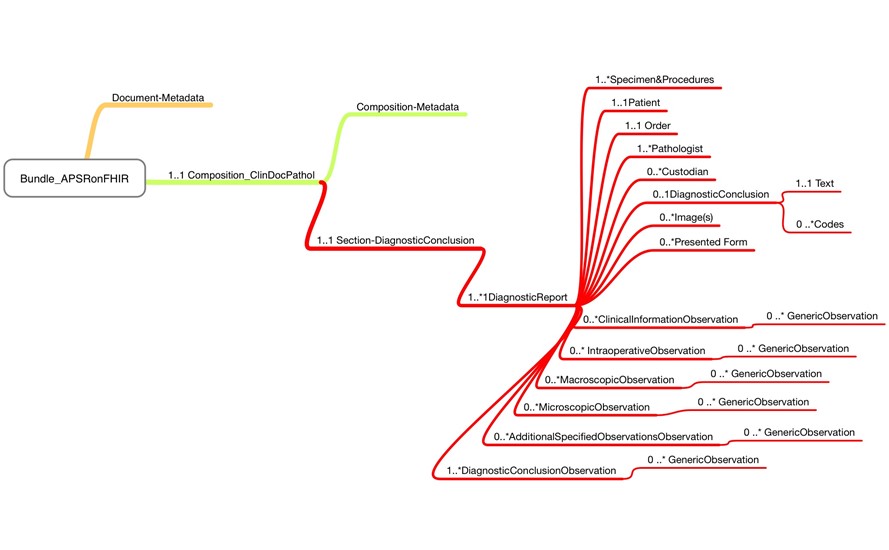
- Link to the current draft: https://simplifier.net/medizininformatikinitiative-modulpathologie
- There is work creating the Diagnostic Report section.
- Diagnostic Conclusion will be a very short observation in the report.
- Group observations to cover the grouping into the CDA sections were created, and then those have all the observations.
- A bundle was created and used composition to support the document. The document will have IG available for the ballot at HL7 Germany in March
- Are there plans to coordinate with SDC for structured data?
- Yes, those structured data are in the individual observations
- Creating a logical model in eXHealth
- Gunter is also the primary author of APSR 2.1 in art décor. He can help support the eXHealth effort, too
- These two efforts might fit together well.
- It was requested that Gunter attends the Tuesday morning meetings.
- OO also came up with the solution of combining composition with DiagnosticReport
- The two approaches should be compared to ensure we can resolve any differences (also include the genomics reporting)
- In Germany, genomics doesn't use composition but does build a similar diagnostic report.
- It should be determined to find a project to make this comparison soon:
- The PH (Helios) and genomics accelerators at HL7 may be the best home for this.
- JD will add awareness for the genomics quarter next week.
- However, we may need a separate project
- The two approaches should be compared to ensure we can resolve any differences (also include the genomics reporting)
- Xe-health
- Two variant options to express the lab content?
- 4/5 countries only using variant 1;
- What was the reasoning for having two variants?
- We allowed this to accommodate the various pdf printed reports - in France; we had two possible layouts:
- French extension of XD-Lab is here: ADD LINK!!!
- variant 1 = single chapter with all results
- variant two = only organizes the narrative blocks by creating a high-level section (Laboratory Speciality section) with subsections (laboratory Report Item)
- The issue is that the example uses a code for a single lab test in the subsection rather than a subsection code, causing some confusion
- It may be worth revising the examples to make it clearer to use a section code here instead.
- The next step is for Juergen to write the CP.
- Two variant options to express the lab content?
- The list of LOINC section codes will need to be updated
- It must be understood that these are example codes and not a complete list.
- ArtDecor implemented this as SHALL.
- Do we also want to allow other code systems?
- SHOULD can have two value set bindings
- Extensible = must be LOINC but can add more codes
- Preferred = could be this or any other code system
- We need to increase the content and make the value set extensible.
- This does not need to track the type of laboratory that created these results, so rename the section to Lab test type rather than lab specialty
- The next step is for Juergen to write the CP to make the value set extensible and change the name from Specialty to Study Type. Juergen will write the CP
- Juergen will have Francois and some others review the above CPs
- Status codes:
- There are three levels:
- Status of observation:
- There is a need for at least: preliminary, completed, aborted, corrected
- XD-lab is intended to support ONLY the communications when the workflow is complete, so preliminary is not needed in that case.
- The group should think about correcting and entered-in-error.
- Entered-in-error:
- It seems to be more of an annotation on the electronic record in the dB.
- The laboratory should be able to reconstruct the history of everything that has been shared, but it is not easy on the consumer side to do with this.
- Corrected:
- What if you send results in different formats? How do you track corrections across various means, and what status should be assigned to each.
- Entered-in-error:
- Status of order:
- Status of document:
- It should not be computed from the status of the observations.
- Juergen will share the discussion around this document
- Riki will share minutes from Lab calls (Fridays 1 – 2 PM ET on OO meeting coordinates) when we are discussing statuses - in general, meeting notes are here – Click on LAB: https://confluence.hl7.org/display/OO/OO+Meeting+Minutes
- Using CDA for workflow?
- CDA is expected to be a snapshot of the data at a specific point in time; CDA can point to the previous report that this report replaces
- CDA is not made to handle workflows - IHE created XDW to address some of these issues.
- France will create a CDA to document orders to a data repository, but not for actual order workflow handling.
- Cross-border e-prescription use is acceptable because you share a persisted prescription document (think of it as a copy) of only those who still have open prescriptions.
- They are updated by providing a new version, but the dispensing status is not part of that new version.
- Status of observation:
- There are three levels:
- We will continue the next IHE PaLM call on Feb 9