PaLM Conf Minutes 2021-June-09
Jump to navigation
Jump to search
Attendees
| Name | |
|---|---|
| Markus Herrmann | |
| Kenichi Takahashi | |
| Alessandro Sulis | Allesandro.sulis@crs4.it |
| Dan Rutz | drutz@epic.com |
| Raj Dash | r.dash@duke.edu |
| Megumi Kondo | megumi.kondo.sakura.japan@gmail.com |
| Ralf Herzog | ralf.herzog@roche.com |
| Francesca Frexia | Francesca.frexia@crs4.it |
| Nick Haarselhorst | nick.haarselhorst@philips.com |
| Mary Kennedy | mkenned@cap.org |
| Gunter Haroske | haroske@icloud.com |
| David de Mena | |
| Gianluca Pavan | Arsenal.IT |
| Alex Goel | agoel@cap.org |
| David Beckman | dBeckman@epic.com |
| Sujith Nair | snair@acr.org |
| Kevin Schap | kschap@cap.org |
| Will Humphrey |
Next meeting: June 15, 2021 with DICOM WG26
Discussion:
- IHE F2F in Ghent Belgium – Tentative dates are September 8-10
- Depends on COVID restrictions
- Meeting request from Digital Pathology = ESDIP
- Meet with DICOM on June 15th 3 – 5 PM Central European time
- Agenda topics to include:
- DPIA
- DICOM annotation work
- Agenda topics to include:
- Meet with DICOM on June 15th 3 – 5 PM Central European time
- DPIA LAB-80 and container ID discussion at DICOM joint meeting
- Discussed at DICOM WG 22
- Two aspects: most of current use case have same specimen and container ID
- HL7 supports separate IDs; how will LIS support this?
- DICOM and HL7 both allow different values for them
- Should provide capability of both
- LIS do not have separate specimen and container ID (e.g., tube of blood)
- DICOM says can use both but took over HL7
- Important to have the capability in DPIA
- Need to establish a real-world use for this
- Will be discussed at joint meeting with DICOM on June 15
- Specimen is a special type of container per Raj
- Dan – has not seen specimen and containers different; there is a container of container in a storage system; unclear of answer; difficult in real world to do
- Specimen ID should be considered a type of container per Raj
- An additional aspect that DICOM WG-26 would like to discuss in joint meeting, is the selection of schemes/systems and value sets for coding anatomical concepts and pathological findings.
- Topics for DICOM joint meeting
- 1. Transferring an annotation back and forth
- 2. Highlighting the annotation / region of interest to be reviewed
- 3. DPIA LAB-80 and Container ID discussion
- RadPath reporting update
- Progress being made on use cases
- Transactions: 1. Form filler to creator; 2. Send concordance report 3. Retrieve concordance report
- Transactions will be done in FHIR
- Creating a possible library of measures
- Form submission – might be removed because adds a lot of complexity
- Should specify form submission? Or leave it open to implementors
- Form submission looks at capturing the data to calculate the measure might not need to be in scope; assume the system has the data; just doing the assessment of the concordance
- Will put in SDC and APSR considerations and will put in a description of how to capture data using either of those
- In profile - Agnostic to how data was captured if you can put the data in FHIR observation
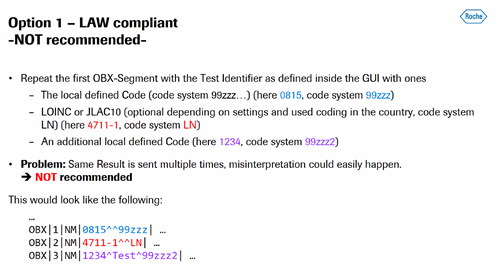
- LAW discussion
- Issue: Up to now the IHE LAW profile is not allowing the sending of Alternate Identifiers together with normal Identifiers while LOINC is recommending it: “The OBX-3 slot allows 2 sets of triplets, one for your local concept and one for the concept from the vocabulary standards.”
- 3 options presented by Ralf:
- Discussion:
- Do we want to have alternate IDs?
- Alessandro – for 2 and 3 there could be cases for both
- Ralf prefers #2 and Alessandro agrees
- Ralf will put through a change proposal
- LAW – LIS and instrument; why need an alternative code or may not know what a LOINC is. What is the use case? Instruments can have local defined code set and then need to distinguish between local and alternative code.
- SET
- Release date is postponed to June 30; Alessandro will inform Mary J
- There is still time to send comments
- Digital Pathology Aid for Telemedicine -Gianluca https://docs.google.com/document/d/1raJKfBhct20uO0jUk3zJYS3XcSxj77G-iSsiLNTPvbs/edit#heading=h.9b4j8x7u7mts
- Completed chapters and use cases
- Specified and resolved issues from last meeting; (RAD-55 and RAD-69)