PaLM Conf Minutes 2018-Dec-12
Jump to navigation
Jump to search
Back to IHE Pathology and Laboratory Medicine (PaLM) Domain
Back to IHE Pathology and Laboratory Medicine (PaLM)Technical Committee Page
Attendees
| Name | Name |
|---|---|
| Riki Merrick | Alessandro Sulis |
| Raj Dash | Francesca Frexia |
| Francois Macary | Mario Villace |
| David Clunie | Dan Rutz |
| Kevin Schap | Jim Harrison |
| Mary Kennedy | Nick Hasselhorst |
| Ian Gabriel | David Beckman |
| Megumi Kondo | Filip Migom |
| Mikael Wintell | |
Minutes
- Francois reviewed the Nov 2018 F2F meeting wrap up and assignments.
- The 2018 PaLM meeting roster will be updated before the DCC December 18 call.
- Raj is working on the Digital Pathology profile and white paper and will finalize the decisions suggested at the F2F.
- The LCC will be sent for final review by the end of the week. It will then be sent to Mary Jungers to publish for comments.
- The next F2F will be in Tokyo, probably May 27 – 29, 2019.
- The call for new proposals is open now; new proposals will be selected in January 2019. Send new proposals to Riki and Francois.
- TMA update: Per Filip, there are implementation projects waiting for this to be published. The meeting between Filip and Epic has been delayed until February 2019. Per Dan, he is working on the updates and intends to have it published in December 2018. It is possible it might be ready in time for the European Connectathon in April (registration is open now and closes the second week in January). Dan stated that if only the validation of message profiles is needed, and we have both partners available, it might be doable to get it into the European Connectathon. Alessandro might be able to help build the test scenario.
- It was brought to the group’s attention that the abbreviations for LCC and TMA are used elsewhere and have publications with those acronyms: LCC (Life Cycle Costs) and TMA (Tumor Micro Array). Francois pointed out that LCC and TMA have been used for over a year now. It was suggested that we refer to them using IHE-LCC and IHE-TMA. Francois said PaLM will consider the suggestion.
- Riki will send a heads up to Mary Jungers about IHE-TMA > IHE-LCC publication
- Francois reviewed the 2019 cycle milestone dates: https://wiki.ihe.net/index.php/Domain_Milestone_Dates
- PaLM will have 4 new supplements for public comment:
- DP-Image acquisition
- DP-ordering-reporting
- LSH
- SET
- PaLM will re-publish 1 (IHE-LCC for public comment now and then as a supplement in March 2019)
- PaLM will have one white paper: digital pathology
- We will also re-publish the TF (we have 1 CP) in the July 1 timeframe.
- PaLM will have 4 new supplements for public comment:
Digital Pathology white paper/profiles update (Raj)
- Talking only about image acquisition profile
- The plan is to finalize this supplement and request publication on March 14. There will be one month for public comment. The intent is that the trial implementation version be prepared for a last review at the Tokyo face to face meeting, end of May, so as to send it for publication right after that meeting.
- DICOM WG26 needs the draft document with clean specification of the HL7 transactions and then they will work on the DICOM transactions.
- DICOM WG26 requests for a non-ambiguous mapping between DICOM data elements and HL7 data elements. This is already done by Radiology domain for patient and order. PaLM will add the specimen data elements, checking what was already done in the context of the legacy APW profile.
- Riki will share the mapping for the DICOM Supplement 122 elements to HL7 specimen DAM – will still need to translate that to HL7v2 – there are a few gaps; this should be done ASAP
- Riki will contact Mikael and Nick as primary contacts and cc David Clunie
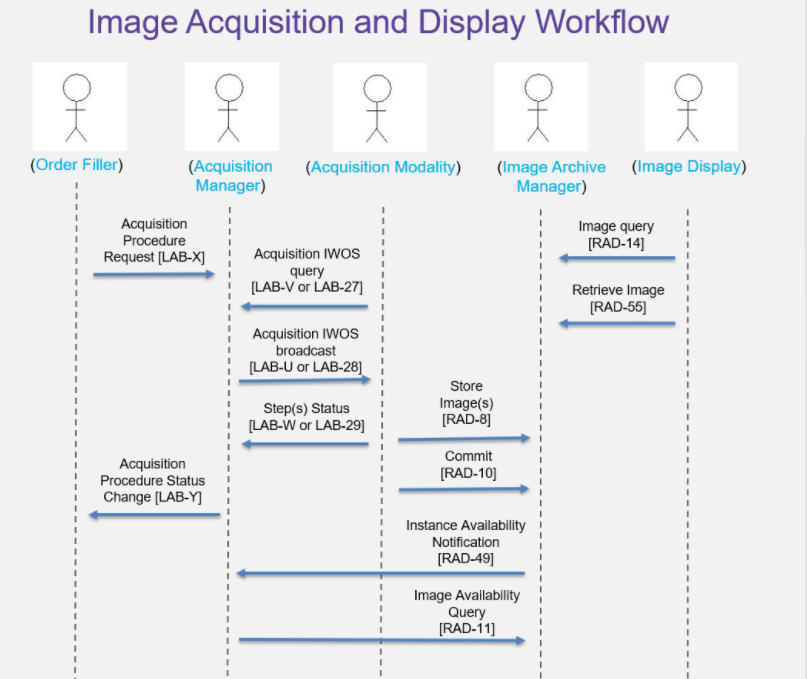
- Regarding the diagram below: Per DICOM, do not worry about the number of transactions on the righthand side of image acquisition modality = DICOM WG 26 will take of those and review what is there
- Create an image ordering profile for order filler to acquisition manager separate from the acquisition workflow profile will work for acquisition modality vendors
- Regarding the profile:
- Include the ppt with the reference figures for the document (and only those
- Need to remove extraneous information – Francois and Riki will clean it up and then share to DICOM WG26
- PaLM will hand the draft profile of imaging acquisition over to WG26 on Monday December 17.
- The DICOM WG26 IHE review group meets every second Tuesday and will review this IHE profile. The next meeting of this group is 12/18 (this is the third Tuesday)
- Raj will distribute the white paper draft by end of next week
- Regarding the profile:
SET update (Alessandro)
- The CR has been divided into the different use cases:
- #1: Specimen collection and specimen container preparation
- Need to decide how many trigger events to define so we can replace Zxx with the appropriate numbers
- Anything related to specimen collection should be in the same structure (collection, missed reason, correction etc.)
- Use EVN-4 reason to indicate why the event is being sent
- Having a separate message for container prep seems reasonable; may need to review if identifiers on labels are used prior to collection here so may need to look at the required fields in SPM segment
- Combine the first two messages
- #2: Specimen movement tracking (specimen centric)
- At the F2F we suggested a single message structure and use the trigger event to identify the situations – similar to ADT messaging approach
- How many workflow steps?
- Arrive = specimen comes in the door
- There is separate step to see, if specimen is acceptable
- Get discussion from different labs what events we need to create in the HL7 WG and have a list of ALL the events that would be needed here (either use the trigger events or EVN)
- Consider where the specimen comes from (if internally or externally may be different) and just specimen accepted cold imply specimen arrived, so may be organization dependent
- Using 2 PRT segments – one to indicate the arriving location and one to indicate the departing location, if needed
- Per Francois, keep four trigger events and keep one message structure.
- SHP is not mandatory? Ask Alessandro.
- #3: Specimen movement tracking (package centric)
- Similar to the specimen movement tracking, but centered on the package, not the specimen – here the SHP and PAC are mandatory
- Need both a shipment and package structure; includes acceptance and rejection of specimens in the same package;
- provide guidance on when to use specimen centric or package centric structure when accepting/rejecting specimens in a package. Ask HL7 WG about preferred approach for a partial rejection (e.g., Package 1 with 10 specimen, there can be 2 messages – one for 8 of the 10 accepted and one message for rejection of 2 of the 10 specimen)
- #4 Specimen Identification tracking
- The structure is good
- #5 Specimen management in biobanks tracking
- Archive and retrieve might be separate use cases
- Specimen discard could be part of specimen reject/disposal; Specimen discarding should be more general – should this just be specimen not available? Container broken (reject reason) or lack of quantity no need to store/ archive
- An example: when I retrieve a specimen from the biobank and then the test corrupts the specimen, it is no longer able to archive; the specimen is not available for the function the specimen is requested
- Specimen rejection can be test specific, while when the specimen is disposed of, then no function will be able to use it
- Maybe we should rename as specimen disposition – either archive and retrieval or disposal (FINAL)
- Name the trigger events as generic as possible, and include some usage notes describing some of the examples for each of the events
- #6 Specimen derivation tracking
- Block to slides / aliquots
- Add an explanation as to why we have the use case grouping in this document, but name the trigger events generic
- #7 Specimen processing tracking
- Riki will try to get on the OO agenda for the WGM in January.
- Alessandro received an email from Peter O Toole (mTuitive) stating that they are interested in the SET profile; Mary Kennedy and invite him to join the group.