PCC TF-1
Volume 1
HIMSS and RSNA
Integrating the Healthcare Enterprise
IHE Patient Care Coordination
Technical Framework
Volume I
Revision 3.0
2008-2009
Public Comment
Preface to Volume 1 of the PCC Technical Framework
Intended Audience
The intended audience of this document is:
- Healthcare professionals involved in informatics
- IT departments of healthcare institutions
- Technical staff of vendors participating in the IHE initiative
- Experts involved in standards development
- Those interested in integrating healthcare information systems and workflows
Related Information for the Reader
The reader of volume 1 should read or be familiar with the following documents:
- Volume 1 of the Cross-Enterprise Document Sharing (XDS) Integration Profile documented in the ITI Infrastructure Technical Framework
(See http://www.ihe.net/Technical_Framework/index.cfm ).
- Volume 1 of the Notification of Document Availability (NAV) Integration Profile documented in the ITI Infrastructure Technical Framework
(See http://www.ihe.net/Technical_Framework/index.cfm ).
- Volume 1 of the Audit Trail and Node Authentication (ATNA) Integration Profile documented in the ITI Infrastructure Technical Framework
(See http://www.ihe.net/Technical_Framework/index.cfm ).
- HL7 Clinical Document Architecture Release 2: Section 1, CDA Overview.
- Care Record Summary – Implementation Guide for CDA Release 2 (US Realm): Section 1
- Presentations from IHE Workshop: Effective Integration of the Enterprise and the Health System - June 28–29, 2005: http://www.ihe.net/Participation/workshop_2005.cfm, June 2005:
- Leveraging IHE to Build RHIO Interoperability
- Cross-Enterprise Document Sharing (XDS)
- Notification of Document Availability (NAV)
- Patient Care Coordination
- Use Cases for Medical Summaries
- Patient Care Coordination - Overview of Profiles
How this Volume is Organized
Section 2 describes the general nature, structure, purpose and function of the Technical Framework. Section 3 and the subsequent sections of this volume provide detailed documentation on each integration profile, including the Patient Care Coordination problem it is intended to address and the IHE actors and transactions it comprises.
The appendices following the main body of the document provide a summary list of the actors and transactions, detailed discussion of specific issues related to the integration profiles and a glossary of terms and acronyms used.
Conventions Used in this Document
This document has adopted the following conventions for representing the framework concepts and specifying how the standards upon which the IHE Technical Framework is based should be applied.
Technical Framework Cross-references
When references are made to another section within a Technical Framework volume, a section number is used by itself. When references are made to other volumes or to a Technical Framework in another domain, the following format is used:
<domain designator> TF-<volume number>: <section number>
where:
- <domain designator>
- is a short designator for the IHE domain (PCC= Patient Care Coordination, ITI = IT Infrastructure, RAD = Radiology)
- <volume number>
- is the applicable volume within the given Domain Technical Framework (e.g., 1, 2, 3), and
- <section number>
- is the applicable section number.
For example: PCC TF-1: 3.1 refers to Section 3.1 in volume 1 of the IHE Patient Care Coordination Technical Framework, ITI TF-2: 4.33 refers to Section 4.33 in volume 2 of the IHE IT Infrastructure Technical Framework.
IHE Actor and Transaction Diagrams and Tables
Each integration profile is a representation of a real-world capability that is supported by a set of actors that interact through transactions. Actors are information systems or components of information systems that produce, manage, or act on categories of information required by operational activities in the enterprise. Transactions are interactions between actors that communicate the required information through standards-based messages.
The diagrams and tables of actors and transactions in subsequent sections indicate which transactions each actor in a given profile must support.
The transactions shown on the diagrams are identified both by their name and the transaction number as defined in PCC TF-2 (Volume 2 of the PCC Technical framework). The transaction numbers are shown on the diagrams as bracketed numbers prefixed with the specific Technical Framework domain.
In some cases, a profile is dependent on a prerequisite profile in order to function properly and be useful. For example, Cross-Enterprise Sharing of Medical Summaries depends on Audit Trail and Node Authentication (ATNA). These dependencies can be found by locating the desired profile in the dependencies section of this document to determine which profile(s) are listed as prerequisites. An actor must implement all required transactions in the prerequisite profiles in addition to those in the desired profile.
Process Flow Diagrams
The descriptions of integration profiles that follow include process flow diagrams that illustrate how the profile functions as a sequence of transactions between relevant actors.
These diagrams are intended to provide an overview so the transactions can be seen in the context of an institution’s or cross-institutions’ workflow. Certain transactions and activities not defined in detail by IHE are shown in these diagrams in italics to provide additional context on where the relevant IHE transactions fit into the broader scheme of healthcare information systems. These diagrams are not intended to present the only possible scenario. Often other actor groupings are possible, and transactions from other profiles may be interspersed.
In some cases the sequence of transactions may be flexible. Where this is the case there will generally be a note pointing out the possibility of variations. Transactions are shown as arrows oriented according to the flow of the primary information handled by the transaction and not necessarily the initiator.
Copyright Permissions
Health Level Seven, Inc., has granted permission to the IHE to reproduce tables from the HL7 standard. The HL7 tables in this document are copyrighted by Health Level Seven, Inc. All rights reserved. Material drawn from these documents is credited where used.
IHE has been very fortunate in having the American College of Obstetricians and Gynecologists (ACOG) help us in the definition of the data found in the Antepartum Summary Profile (APS).
The Antepartum Summary Profile (APS) describes the content structures and specifications the American College of Obstetricians and Gynecologists (ACOG) views are necessary in an antepartum record. ACOG encourages the use of the content structures contained in the Antepartum Summary Profile of the Patient Care Coordination Technical Framework. ACOG does not endorse any EMR products. Companies or individuals that use these content structures in EMR product or service are prohibited from using ACOG's name and/or its logo on any promotional material, packaging, advertisement, website or in any other context related to the EMR product or service.
Braden Scale For Predicting Pressure Sore Risk, Copyright © Barbara Braden and Nancy Bergstrom, 1988. Reprinted with permission. Barabara Braden and Nancy Bergstrom have granted permission to use the Braden Scale in the IHE Functional Status Assessment Integration Profile to be provided to vendors for demonstration purposes only. Should a vendor chose to include the Braden Scale in their product, they must seek permission to do so from the copyright holders. More information is available from http://www.bradenscale.com/
Introduction
This document, the IHE Patient Care Coordination Technical Framework (PCC TF), defines specific implementations of established standards. These are intended to achieve integration goals that promote appropriate exchange of medical information to coordinate the optimal patient care among care providers in different care settings. It is expanded annually, after a period of public review, and maintained regularly through the identification and correction of errata. The latest version of the document is always available via the Internet at http://www.ihe.net/Technical_Framework/ , where the technical framework volumes specific to the various healthcare domains addressed by IHE may be found.
The IHE Patient Care Coordination Technical Framework identifies a subset of the functional components of the healthcare enterprises and health information networks, called IHE actors, and specifies their interactions in terms of a set of coordinated, standards-based transactions. The other domains within the IHE initiative also produce Technical Frameworks within their respective areas that together form the IHE Technical Framework. Currently, the following IHE Technical Framework(s) are available:
- IHE IT Infrastructure Technical Framework
- IHE Cardiology Technical Framework
- IHE Laboratory Technical framework
- IHE Radiology Technical Framework
- IHE Patient Care Coordination Technical Framework
Where applicable, references are made to other technical frameworks. For the conventions on referencing other frameworks, see the preface of this volume.
Relationship to Standards
The IHE Technical Framework identifies functional components of a distributed healthcare environment (referred to as IHE actors), solely from the point of view of their interactions in the healthcare enterprise. It further defines a coordinated set of transactions based on standards (such as HL7, IETF, ASTM, DICOM, ISO, OASIS, etc.) in order to accomplish a particular use case. As the scope of the IHE initiative expands, transactions based on other standards may be included as required.
At its current level of development, IHE has also created Content Integration Profiles to further specify the payloads of these transactions, again based on standards. This has become necessary as the healthcare industry moves towards the use of transaction standards that have been used in more traditional computing environments.
In some cases, IHE recommends selection of specific options supported by these standards. However, IHE does not introduce technical choices that contradict conformance to these standards. If errors in or extensions to existing standards are identified, IHE’s policy is to report them to the appropriate standards bodies for resolution within their conformance and standards evolution strategy.
IHE is therefore an implementation framework, not a standard. Conformance claims for products must still be made in direct reference to specific standards. In addition, vendors who have implemented IHE integration capabilities in their products may publish IHE Integration Statements to communicate their products’ capabilities. Vendors publishing IHE Integration Statements accept full responsibility for their content. By comparing the IHE Integration Statements from different products, a user familiar with the IHE concepts of actors and integration profiles can determine the level of integration between them. See PCC TF-1: Appendix C for the format of IHE Integration Statements.
Relationship to Product Implementations
The IHE actors and transactions described in the IHE Technical Framework are abstractions of the real-world healthcare information system environment. While some of the transactions are traditionally performed by specific product categories (e.g. HIS, Clinical Data Repository, Electronic Health record systems, Radiology Information Systems, Clinical Information Systems or Cardiology Information Systems), the IHE Technical Framework intentionally avoids associating functions or actors with such product categories. For each actor, the IHE Technical Framework defines only those functions associated with integrating information systems. The IHE definition of an actor should therefore not be taken as the complete definition of any product that might implement it, nor should the framework itself be taken to comprehensively describe the architecture of a healthcare information system.
The reason for defining actors and transactions is to provide a basis for defining the interactions among functional components of the healthcare information system environment. In situations where a single physical product implements multiple functions, only the interfaces between the product and external functions in the environment are considered to be significant by the IHE initiative. Therefore, the IHE initiative takes no position as to the relative merits of an integrated environment based on a single, all-encompassing information system versus one based on multiple systems that together achieve the same end.
Framework Development and Maintenance
The IHE Patient Care Coordination Technical Framework is continuously maintained and expanded on an annual basis by the IHE Patient Care Coordination Technical Committee. The development and maintenance process of the Framework follows a number of principles to ensure stability of the specification so that both vendors and users may use it reliably in specifying, developing and acquiring systems with IHE integration capabilities.
The first of these principles is that any extensions or clarifications to the Technical Framework must maintain backward compatibility with previous versions of the framework (except in rare cases for corrections) in order to maintain interoperability with systems that have implemented IHE Actors and Integration Profiles defined there. The IHE Patient Care Coordination Technical Framework is developed and re-published annually following a three-step process:
- The Patient Care Coordination Technical Committee develops supplements to the current stable version of the Technical Framework to support new functionality identified by the IHE Strategic and PCC Planning Committees and issues them for public comment.
- The Committee addresses all comments received during the public comment period and publishes an updated version of the Technical Framework for “Trial Implementation.” This version contains both the stable body of the Technical Framework from the preceding cycle and the newly developed supplements. It is this version of the Technical Framework that is used by vendors in developing trial implementation software for the IHE Connectathons.
- The Committee regularly considers change proposals to the Trial Implementation version of the Technical Framework, including those from implementers who participate in the Connectathon. After resolution of all change proposals received within 60 days of the Connectathon, the Technical Framework version is published as “Final Text”.
As part of the Technical framework maintenance the Committee will consider change proposals received after the publication to the “Final Text”.
About the Patient Care Coordination Integration Profiles
IHE Integration Profiles offer a common language that healthcare professionals and vendors can use to discuss integration needs of healthcare enterprises and the integration capabilities of information systems in precise terms. Integration Profiles specify implementations of standards that are designed to meet identified clinical needs. They enable users and vendors to state which IHE capabilities they require or provide, by reference to the detailed specifications of the IHE Patient Care Coordination Technical Framework.
Integration profiles are defined in terms of IHE Actors, transactions and their content. Actors (listed in PCC TF-1: Appendix A) are information systems or components of information systems that produce, manage, or act on information associated with clinical and operational activities. Transactions (listed in PCC TF-1: Appendix B) are interactions between actors that communicate the required information through standards-based messages. Content is what is exchanged in these transactions, and are defined by Content Profiles.
Vendor products support an Integration Profile by implementing the appropriate actor(s) and transactions. A given product may implement more than one actor and more than one integration profile.
Content Profiles define how the content used in a transaction is structured. Each transaction is viewed as having two components, a payload, which is the bulk of the information being carried, and metadata that describes that payload. The binding of the Content to an IHE transaction specifies how this payload influences the metadata of the transaction. Content modules within the Content Profile then define the payloads. Content modules are transaction neutral, in that what they describe is independent of the transaction in which they are used, whereas content bindings explain how the payload influences the transaction metadata.
The figure below shows the relations between the Content Integration Profiles of the Patient Care Coordination Domain.
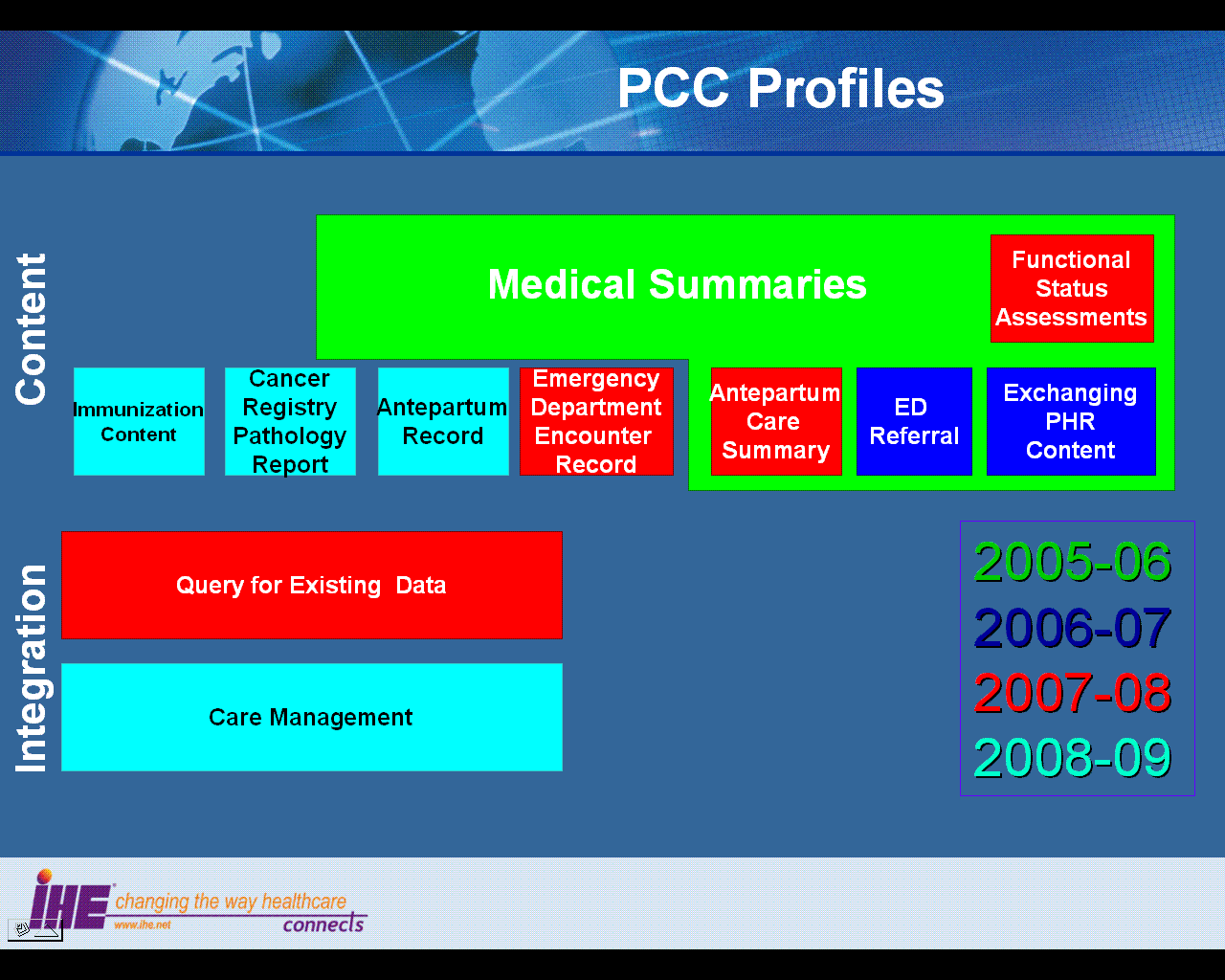
Dependencies of the PCC Integration Profiles
Dependencies among IHE Integration Profiles exist when implementation of one integration profile is a prerequisite for achieving the functionality defined in another integration profile. The table below defines these dependencies. Some dependencies require that an actor supporting one profile be grouped with one or more actors supporting other integration profiles. For example, Cross-Enterprise Sharing of Medical Summaries (XDS-MS) requires that its actors be grouped with a Secured Node Actor of the Audit Trail and Node Authentication (ATNA) Integration Profile. The dependency exists because XDS-MS and XDS actors must support a secured communication channel with proper auditing of the exchange of patient identified information in order to function properly in an environment where protection of patient privacy is critical.
| Integration Profile | Depends on | Dependency Type | Purpose |
| All PCC Content Profiles |
Audit Trail and Node Authentication (ATNA) | Each Content Creator and Content Consumer actor shall be grouped with the ATNA Secured Node Actor | Required to manage audit trail of exported PHI, node authentication, and transport encryption. |
| Consistent Time (CT) | Each Content Creator and Content Consumer actor shall be grouped with the Time Client Actor | Required to manage and resolve conflicts in multiple updates. | |
| Functional Status Assessments (FSA) | Cross Enterprise Document Exchange of Medical Summaries (XDS-MS) OR Exchange of Personal Health Record Content (XPHR) OR Emergency Department Referral (EDR) |
Content Consumers implementing the Functional Status Assessments profile shall be grouped with either the XDS-MS, XPHR or EDR Content Consumer. Content Creators implementing the Functional Status Assessments profile shall be grouped with either the XDS-MS, XPHR or EDR Content Creator. | Ensures that the Functional Status Assessment is communicated as part of an exchange of medical summary information. |
| Functional Status Assessments (QED) | Audit Trail and Node Authentication (ATNA) | Each actor in this profile shall be grouped with the ATNA Secure Node or Secure Application actor. | Required to manage audit trail of exported PHI, node authentication, and transport encryption. |
| Consistent Time (CT) | Each actor in this profile shall be grouped with the Time Client Actor | Required to manage and resolve conflicts in multiple updates. |
To support a dependent profile, an actor must implement all required transactions in the prerequisite profiles in addition to those in the dependent profile. In some cases, the prerequisite is that the actor selects any one of a given set of profiles.
PCC Integration Profiles Overview
In this document, each IHE Integration Profile is defined by:
- The IHE actors involved
- The specific set of IHE transactions exchanged by each IHE actor.
- The content of the IHE transactions
These requirements are presented in the form of a table of transactions required for each actor supporting the Integration Profile. Actors supporting multiple Integration Profiles are required to support all the required transactions of each Integration Profile supported. When an Integration Profile depends upon another Integration Profile, the transactions required for the dependent Integration Profile have not been included in the table.
The content of the transactions are presented as Content Integration Profiles. These are specification of the content to be exchange, along with explanations (called bindings) of how the content affects the transactions in which it is exchanged. It is expected that Content Integration Profiles will be used environments where the physician offices and hospitals have a coordinated infrastructure that serves the information sharing needs of this community of care. Several mechanisms are supported by IHE profiles:
- A registry/repository-based infrastructure is defined by the IHE Cross-Enterprise Document Sharing (XDS) and other IHE Integration Profiles such as patient identification (PIX & PDQ), and notification of availability of documents (NAV).
- A media-based infrastructure is defined by the IHE Cross-Enterprise Document Media Interchange (XDM) profile.
- A reliable messaging-based infrastructure is defined by the IHE Cross-Enterprise Document Reliable Interchange (XDR) profile.
- All of these infrastructures support Security and privacy through the use of the Consistent Time (CT) and Audit Trail and Node Authentication (ATNA) profiles.
For more details on these profiles, see the IHE IT Infrastructure Technical Framework, found here: http://www.ihe.net/Technical_Framework/.
Such an infrastructure is assumed by the use cases that focus on the context for defining the specific clinical information content for this profile. These content integration profiles use similar transactions and differ only in the content exchanged. A process flow for these use cases using Cross Enterprise Document Sharing (XDS) and Notification of Document Availability (NAV) is shown in the figure below. Other process flows are possible using XDM and/or XDR.

These steps are:
- Extract/capture a collection of records into a set of documents packaged as an XDS Submission Set. This submission contains at least one clinical document, and may contain a number of other related clinical documents. For example, Medical Summaries are clinical documents (already known in the paper world), which often serve the dual purpose of documenting an encounter and providing the rationale for sending the information to another provider. This step utilizes the transactions provided by the ITI XDS profile to place the records in an XDS Repository (local or shared).
- The Repository ensures that the documents of the submission set are registered with the XDS Registry of the Affinity Domain (set of cooperating care delivery institutions).
- Notify the other provider that documents are now available for review. This step utilizes the transactions provided by the ITI NAV profile to perform the e-mail notification.
- The e-mail notification that contains no patient identified information is received by the specialist EMR system.
- The receiving provider can then utilize existing query transactions from the XDS profile to find the URL of the Documents.
- Finally, the receiving provider may choose to display the document, or import relevant information from these records into their own EMR system.
Unplanned Access to past Content
In many cases, a provider may need to assess information from the patient care history, and patients may have content in the XDS repository from prior visits to other providers. For example, Medical Summaries, as well as other documents such as laboratory and radiology reports are critical for emergency physicians and nurses to provide the best care to patient in acute conditions. The figure below shows the transactions required for this use case, again, using XDS. Other process flows are possible using XDM and/or XDR.
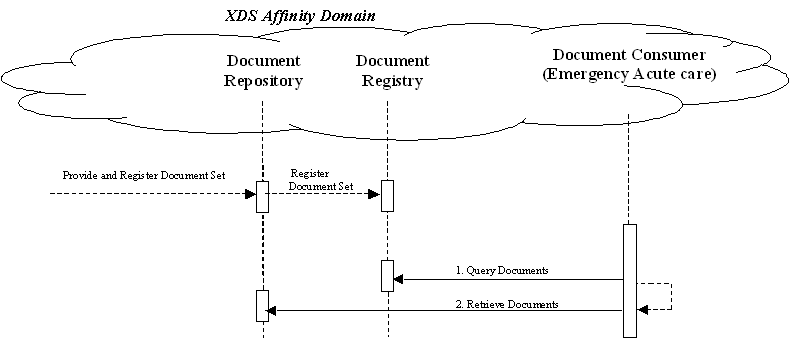
Note that IHE Integration Profiles are not statements of conformance to standards, and IHE is not a certifying body. Users should continue to request that vendors provide statements of their conformance to standards issued by relevant standards bodies, such as HL7 and DICOM. Standards conformance is a prerequisite for vendors adopting IHE Integration Profiles.
Also note that there are critical requirements for any successful integration project that IHE cannot address. Successfully integrating systems still requires a project plan that minimizes disruptions and describes fail-safe strategies, specific and mutually understood performance expectations, well-defined user interface requirements, clearly identified systems limitations, detailed cost objectives, plans for maintenance and support, etc.
History of Annual Changes
In the 2005-2006 cycle of the IHE Patient Care Coordination initiative, the first release of the IHE PCC Technical Framework introduced the following integration profile:
- Cross-Enterprise Sharing of Medical Summaries (XDS-MS) – a mechanism to automate the sharing process between care providers of Medical Summaries, a class of clinical documents that contain the most relevant portions of information about the patient intended for a specific provider or a broad range of potential providers in different settings. Medical Summaries are commonly created and consumed at points in time of transfers of care such as referrals or discharge.
In the 2006-2007 cycle of the IHE Patient Care Coordination initiative, the following integration profiles were added to the technical framework.
- Exchange of Personal Health Record Content (XPHR) – provides a standards-based specification for managing the interchange of documents between a Personal Health Record used by a patient and systems used by other healthcare providers to enable better interoperability between these systems.
- Basic Patient Privacy Consents (BPPC) – enables XDS Affinity Domains to be more flexible in the privacy policies that they support, by providing mechanisms to record patient privacy consents, enforce these consents, and create Affinity Domain defined consent vocabularies that identify information sharing policies.
Please Note: This profile was transfered to the ITI Domain in the Fall of 2007, and can be found here http://www.ihe.net/Technical_Framework/index.cfm#IT - Preprocedure History and Physical Content Profile (PPHP) – supports the exchange of information allowing for the assessment and amelioration of risks related to a procedure. Please Note: This profile has been withdrawn.
- Emergency Department Referral Profile (EDR) – provides a means to communicate medical summary data from an EHR System to an EDIS System.
In the 2007-2008 cycle of the IHE Patient Care Coordination initiative, the following integration profiles were added to the technical framework.
- Antepartum Care Summary (APS) - describes the content and format of summary documents used during antepartum care.
- Emergency Department Encounter Summary (EDES) - describes the content and format of records created during an emergency department visit.
- Functional Status Assessment Profile (FSA) - supports the handoff of assessment information between practictioners during transfers of care by defining the Functional Status Assessment option on the XDS-MS and XPHR profiles.
- Query for Existing Data (QED) - allows information systems to query data repositories for clinical information on vital signs, problems, medications, immunizations, and diagnostic results.
- Public Health Laboratory Report(PHLAB) - extends the XD*-LAB profile to support reporting from public health laboratories for disease surviellance activities. Please Note: This profile has been subsequently moved to the XD-LAB specification, and can be found here http://www.ihe.net/Technical_Framework/index.cfm#LAB
In addition, all content within the technical framework was revised in the 2007-2008 cycle to encourage compatibility with the ASTM/HL7 Continuity of Care Document Implementation Guide.
In the 2008-2009 cycle of the IHE Patient Care Coordination initiative, the following integration profiles were added to the technical framework.
- Antepartum Record (APR) - describes the content and format of summary documents used during antepartum care.
- Care Management (CM) - describes the content and format of summary documents used during antepartum care.
- Immunization Content (IC) - describes the content and format of summary documents used during antepartum care.
![]() -->>Care Management and Immunization Content text <<--
-->>Care Management and Immunization Content text <<--
Product Implementations
Developers have a number of options in implementing IHE actors and transactions in product implementations. The decisions cover three classes of optionality:
- For a system, select which actors it will incorporate (multiple actors per system are acceptable).
- For each actor, select the integration profiles in which it will participate.
- For each actor and profile, select which options will be implemented.
All required transactions must be implemented for the profile to be supported (for XDS-MS, refer to the transaction descriptions for XDS in ITI TF-2).
Implementers should provide a statement describing which IHE actors, IHE integration profiles and options are incorporated in a given product. The recommended form for such a statement is defined in PCC TF-1: Appendix C.
In general, a product implementation may incorporate any single actor or combination of actors. When two or more actors are grouped together, internal communication between actors is assumed to be sufficient to allow the necessary information flow to support their functionality; for example, the Document Source Actor of XDS-MS may use the Patient Identifier Cross-reference Consumer Actor to obtain the necessary patient identifier mapping information from its local patient id to that used in the document sharing domain. The exact mechanisms of such internal communication are outside the scope of the IHE Technical Framework.
When multiple actors are grouped in a single product implementation, all transactions originating or terminating with each of the supported actors shall be supported (i.e., the IHE transactions shall be offered on an external product interface).
The following examples describe which actors typical systems might be expected to support. This is not intended to be a requirement, but rather to provide illustrative examples.
An acute care EMR serving a hospital might include a Document Source Actor, Document Consumer Actor, a Document Repository Actor, a Patient Identification Consumer Actor, as well as a Secured Node Actor. An Ambulatory EMR serving a physician practice might include a Document Source Actor, Document Consumer Actor, a Patient Demographics Client Actor, as well as a Secured Node Actor.
Patient Care Coordination Integration Profiles
- (PCC TF-1/EDR)Emergency Department Referral (EDR) Integration Profile
- (PCC TF-1/APS)Antepartum Summary Integration Profile
- (PCC TF-1/FSA)Functional Status Assessments (FSA) Integration Profile
- (PCC TF-1/QED)Query for Existing Data (QED) Integration Profile
- (PCC TF-1/APR)Antepartum Record (APR) Integration Profile
- (PCC TF-1/CM)Care Management (CM) Integration Profile
- (PCC TF-1/IC)Immunization Content (IC) Integration Profile
Appendix A - Actor Descriptions
Actors are information systems or components of information systems that produce, manage, or act on information associated with operational activities in the enterprise.
- Content Creator
- The Content Creator Actor is responsible for the creation of content and transmission to a Content Consumer.
- Content Consumer
- A Content Consumer Actor is responsible for viewing, import, or other processing of content created by a Content Creator Actor.
- Clinical Data Consumer
- A clinical data consumer makes use of clinical patient data.
- Clinical Data Source
- A Clinical Data Sources maintains patient information about vital signs, problem and allergies, results from diagnostic tests (e.g., Lab, Imaging, or other test results), medications, immunizations or historical or planned visits and procedures.
Appendix B - Transaction Descriptions
Transactions are interactions between actors that transfer the required information through standards-based messages. The PCC Technical Framework does not define any specific transactions, as these are assumed to be carried out through the use of transactions defined in other IHE Profiles.
- Query Existing Data
- Request information about recent patient information, used to obtain vital signs measurements, problems and allergies, diagnostic results, medications, immunizations, or procedures or visits relevant for a patient. The query may request information about some or all of the above topics, or may request information on a specific topic, or one entered for a specific encounter or date range.
Appendix C - How to Prepare an IHE Integration Statement
IHE Integration Statements are documents prepared and published by vendors to describe the conformance of their products with the IHE Technical Framework. They identify the specific IHE capabilities a given product supports in terms of IHE actors and integration profiles described in the technical frameworks of each domain.
Users familiar with these concepts can use Integration Statements to determine what level of integration a vendor asserts a product supports with complementary systems and what clinical and operational benefits such integration might provide. Integration Statements are intended to be used in conjunction with statements of conformance to specific standards (e.g., HL7, IETF, DICOM, W3C, etc.).
IHE provides a process for vendors to test their implementations of IHE actors and integration profiles. The IHE testing process, culminating in a multi-party interactive testing event called the Connectathon, provides vendors with valuable feedback and provides a baseline indication of the conformance of their implementations. The process is not intended to independently evaluate, or ensure, product compliance. In publishing the results of the Connectathon and facilitating access to vendors' IHE Integration Statements, IHE and its sponsoring organizations are in no way attesting to the accuracy or validity of any vendor's IHE Integration Statements or any other claims by vendors regarding their products.
IMPORTANT -- PLEASE NOTE: Vendors have sole responsibility for the accuracy and validity of their IHE Integration Statements. Vendors' Integration Statements are made available through IHE simply for consideration by parties seeking information about the integration capabilities of particular products. IHE and its sponsoring organizations have not evaluated or approved any IHE Integration Statement or any related product, and IHE and its sponsoring organizations shall have no liability or responsibility to any party for any claims or damages, whether direct, indirect, incidental or consequential, including but not limited to business interruption and loss of revenue, arising from any use of, or reliance upon, any IHE Integration Statement.
Structure and Content of an IHE Integration Statement
An IHE Integration Statement for a product shall include:
- The Vendor Name
- The Product Name (as used in the commercial context) to which the IHE Integration Statement applies.
- The Product Version to which the IHE Integration Statement applies.
- A publication date and optionally a revision designation for the IHE Integration Statement.
- The following statement: "This product implements all transactions required in the IHE Technical Framework to support the IHE Integration Profiles, Actors and Options listed below:"
- A list of IHE Integration Profiles supported by the product and, for each Integration Profile, a list of IHE Actors supported. For each integration profile/actor combination, one or more of the options defined in the IHE Technical Framework may also be stated. Profiles, Actors and Options shall use the names defined by the IHE Technical Framework Volume I. (Note: The vendor may also elect to indicate the version number of the Technical Framework referenced for each Integration Profile.)
Note that implementation of the integration profile implies implementation of all required transactions for an actor as well as selected options.
The statement shall also include references and/or internet links to the following information:
- Specific internet address (or universal resource locator [URL]) where the vendor's Integration Statements are posted
- URL where the vendor's standards conformance statements (e.g., HL7, DICOM, etc.) relevant to the IHE transactions implemented by the product are posted.
- URL of the IHE Initiative's web page for general IHE information www.himss.org/ihe.
An IHE Integration Statement is not intended to promote or advertise aspects of a product not directly related to its implementation of IHE capabilities.
Format of an IHE Integration Statement
Each Integration Statement shall follow the format shown below. Vendors may add a cover page and any necessary additional information in accordance with their product documentation policies.
| IHE Integration Statement | Date | 12 Oct 2005 |
| Vendor | Product Name | Version |
| Any Medical Systems Co. | IntegrateRecord | V2.3 |
| This product implements all transactions required in the IHE Technical Framework to support the IHE Integration Profiles, Actors and Options listed below: | ||
| Integration Profiles Implemented | Actors Implemented | Options Implemented |
| Cross-Enterprise Sharing of Medical Summaries | Document Consumer | View Option |
| Audit Trail and Node Authentication | Secure Node | none |
| Patient Identity Cross-referencing | Patient Identifier Cross-reference Consumer | PIX Update Notification |
| Internet address for vendor's IHE information:www.anymedicalsystemsco.com/ihe | ||
| Links to Standards Conformance Statements for the Implementation | ||
| HL7 | www.anymedicalsystemsco.com/hl7 | |
| Links to general information on IHE | ||
| In North America: www.ihe.het | In Europe: www.ihe-europe.org | In Japan: www.jira-net.or.jp/ihe-j |
IHE Integration Statement template
An IHE Integration Statement template (MS Word version) is available here.
The IHE Product Registry
The assumption of an integration statement is that all actors listed are functionally grouped and conform to any profile specifications for such groupings. In case of exceptions the vendor must explicitly describe the functional groupings.
IHE has developed a new Web-based database of Integration Statements. The IHE Product Registry enables developers to create, manage and publish Integration Statements for their commercial and open source healthcare IT systems. It allows users to browse for these systems based on their conformance with specific IHE Actors and Profiles. The system is open for use by developers and users now!
Appendix D - Braden Scale for Predicting Pressure Sore Risk
See File:Braden.pdf
Glossary
The following terms are used in various places within this technical framework, and are defined below. The complete IHE Glossary is available on the IHE Wiki at http://wiki.ihe.net/index.php/IHE_Glossary .
- Actor
- An entity within a use case diagram that can perform an action within a use case diagram. Possible actions are creation or consumption of a message
- Acuity Assessment
Also known as triage category, this is the acuity of the patient assigned during the process of ED triage. A number of evidenced based triage scales exist, including the Emergency Severity Index (ESI), Canadian Triage and Acuity Scale (CTAS), the Australasian Triage Scale (ATS), and the Manchester Triage System. In many emergency departments, patients may simply be classified as emergent, urgent or non-urgent.
- ADT
- Admit, Discharge & Transfer.
- Affinity Domain Policy
- Affinity Domain Policy that clearly defines the appropriate uses of the XDS Affinity Domain. Within this policy is a defined set of acceptable use Privacy Consent Policies that are published and understood.
- ASTM
- Formerly the American Society of Testing and Materials, now ASTM International. An SDO that develops a number of standards across a wide variety of industries, including healthcare.
- ATNA
- Audit Trail and Node Authentication. An IHE ITI profile.
- Care Context
- The participations surrounding the care provision act, and the attributes of that act. Everything in the document header. Data history, links to clinical reasoning.
- Continuity of Care Document(CCD)
- An HL7 Clinical Document Architecture (CDA) implementation alternative to ASTM ADJE2369 for institutions or organizations committed to HL7 standards. This specification was developed as a collaborative effort between ASTM and HL7. More information is available from http://www.hl7.org.
- Continuity of Care Record (CCR)
- A core data set of the most relevant administrative, demographic, and clinical information facts about a patient’s healthcare, covering one or more encounters. The CCR is Designation E2369-05 of the ASTM (American Society for Testing and Materials, International). More information is available from http://www.astm.org.
- Clinical Document Architecture (CDA)
- An HL7 standard for the exchange for clinical documents. It specifies the structure and semantics of clinical documents. More information is available from http://www.hl7.org.
- Content Binding
- A content binding describes how the payload used in an IHE transaction is related to and/or constrained by the data elements contained within the content sent or received in those transactions.
- CRS
- Care Record Summary. An implementation guide that constrains CDA Release 2 for Care Record Summary documents.
- CT
- Consistent Time Integration Profile.
- DICOM
- Digital Imaging and Communication in Medicine
- DSG
- Digital Signatures. An IHE ITI Profile.
- EDIS
- An Emergency Department Information System (EDIS) is an extended EHR system used to manage data in support of Emergency Department patient care and operations. The functions of an EDIS may be provided by a single application or multiple applications.
- eMPI
- Enterprise Master Patient Index.
- EMR
- Electronic Medical Record, an Electronic Health Record system used within an enterprise to deliver care (also called EHR-CR by IHE-XDS).
- Estimated Time of Arrival
- the time the patient being referred can be expected to arrive in the emergency department.
- EUA
- Enterprise User Authentication Integration Profile.
- Expected Actions
- Actions which should occur as the result of a trigger event.
- HIMSS
- Healthcare Information and Management Systems Society.
- HL7
- Health Level Seven
- HIS
- Hospital Information System.
- IHE
- Integrating the Healthcare Enterprise.
- Interaction Diagram
- A diagram that depicts data flow and sequencing of events.
- IT
- Information Technology.
- Logical Observation Identifiers Names and Codes (LOINC®)
- A vocabulary developed by the Regenstrief Institute aimed at standardizing laboratory and clinical codes for use in clinical care, outcomes management, and research. Additional information found at http://www.regenstrief.org/medinformatics/loinc/.
- Mode of Arrival
- The method of transportation used to transport the patient to the Emergency Department.
- MPI
- Master Patient Index.
- MRN
- Medical Record Number.
- NAV
- Notification of Document Availability
- OID
- Object Identifier. (See also 'Globally Unique Identifier').
- Patient Identifier Cross-reference Domain
- Consists of a set of Patient Identifier Domains known and managed by a Patient Identifier Cross-reference Manager Actor. The Patient Identifier Cross-reference Manager Actor is responsible for providing lists of "alias" identifiers from different Patient Identifier Domains.
- Patient Identifier Domain
- A single system or a set of interconnected systems that all share a common identification scheme for patients. Such a scheme includes: (1) a single identifier-issuing authority, (2) an assignment process of an identifier to a patient, (3) a permanent record of issued patient identifiers with associated traits, and (4) a maintenance process over time. The goal of Patient Identification is to reduce errors.
- Portable Document Format.
- PIX
- Patient Identifier Cross Referencing. An IHE ITI Profile.
- PDQ
- Patient Demographics Query. An IHE ITI Profile.
- PHR
- Personal Health Record
- Procedure
- In the context of a "Pre-procedure History and Physical," the "procedure" is a surgery or an invasive examination of a patient that is required by quality review organizations to be preceded by a pre-procedure assessment of procedure risk and anesthesia risk. This assessment is typically referred to as a "Pre-operative" or "Pre-procedure History and Physical."
- Process Flow Diagram
- A graphical illustration of the flow of processes and interactions among the actors involved in a particular example.
- Proposed disposition
- the intended disposition (i.e. admission to ICU, discharge to home, transfer to psychiatric hospital), if known, that the referring provider expects the patient will end up after the emergency department intervention.
- Referral Source
- An individual, group, or agency that determined the patient should seek care at the ED. Referral source may be used to determine appropriate discharge referrals and services, or to provide surveillance data for program and service planning, or to examine referral patterns.
- Role
- The actions of an actor in a use case.
- RSNA
- Radiological Society of North America.
- sig.
- A Latin abbreviation for signetur used to represent the instruction following the medication name.
- Scope
- A brief description of the transaction.
SNOMED-CT® A comprehensive clinical terminology, originally created by the College of American Pathologists (CAP) and, as of April 2007, owned, maintained, and distributed by the International Health Terminology Standards Development Organisation (IHTSDO), a non-for-profit association in Denmark. The CAP continues to support SNOMED CT operations under contract to the IHTSDO and provides SNOMED-related products and services as a licensee of the terminology. More information available from http://www.ihtsdo.org/ or the United States National Library of Medicine at http://www.nlm.nih.gov/research/umls/Snomed/snomed_main.html
- Transport Mode
- the method the patient employs, or is provided to get to the emergency department.
- Trigger Event
- An event such as the reception of a message or completion of a process, which causes another action to occur.
- UID
- Unique Identifier (See also Globally Unique Identifier).
- Universal ID
- Unique identifier over time within the UID type. Each UID must belong to one of specifically enumerated species. Universal ID must follow syntactic rules of its scheme.
- Use Case
- A graphical depiction of the actors and operation of a system.
- XUA
- Cross Enterprise User Authentication
- XDS
- Cross Enterprise Document Sharing
