Laboratory Specimen Handoff
Proposed Work Item: Laboratory Specimen Handoff (LSH)
Proposal Editor: John Hopson, Abbott Laboratories
Work Item Editor: John Hopson
Date: 05/22/2015, last updated 05/22/2015
Version: 1.0 (draft)
Domain: Clinical Laboratory
Status: LSH Update Proposal Spring 2015.
Summary
Passing of specimens between Laboratory Automation Systems (LAS) and Specimen Processing Devices (SPD) is an integral part of clinical laboratory operation. In the absence of any industry guidance, such handoffs are currently defined on an ad hoc basis. Implementers naturally turn to predecessor products for guidance. This approach causes similar interfaces to be created within each organization, even though such interfaces share features with products from a variety of vendors.
There clearly exists a need for fully specified interface protocols to reduce the time and costs associated with managing sample handoff in clinical laboratories. The IHE-LAB Committee is the appropriate entity to create and promote such solutions. Prior work by the Committee on the LDA profile is evidence of that need.
This proposed profile will enable LAS and SPD to implement specimen handoff using standardized protocols, reducing the new product design burden for all parties.
The Problem
Current LAS and SPD vendors have little guidance on how to implement specimen handoff interfaces between their equipment. HL7 and CLSI AUTO3-A provide some direction, but do not provide specific guidance for individual handoff workflows.
Use Cases
There are multiple use cases for specimen handoff in the laboratory. The most common is the single-sample, point-in-space presentation use case. The second most common is the single-sample container transfer use case. A third common use case is the passing of a rack with multiple specimen containers between LAS and SPD
This proposal will be implemented in at least three phases, in order of commonality.
- Phase 1 - Single-Sample Point-in-Space Presentation Use Case
LAS notifies the SPD that a specimen container is en route to the IVD analyzer. The SPD prepares for specimen arrival, typically receiving work order steps. Upon arrival, the LAS presents the specimen for aspiration. The SPD acquires the sample from the container. Automation removes the sample from the aspiration position.
- Phase 2 - Single-Sample Container Transfer Use Case
(tbd)
- Phase 3 - Multiple-Sample Container Transfer Use Case
(tbd)
Standards and Systems
Existing systems that could be involved in the solution.
- IVD analyzer
- Specimen processing devices, including IVD analyzers and laboratory pre/post processing devices.
Relevant standards upon which the solution might be based.
- HL7
- CLSI AUTO5-A (HL7 Chapter 13
Technical Approach Overview
Actors
The proposed LDA actors are shown below.
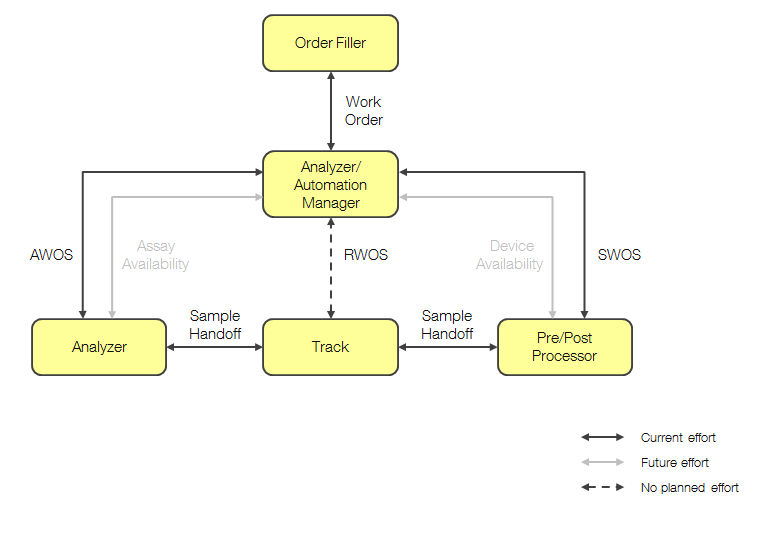
- Order Filler - Receives the laboratory work for each patient
- Analyzer/Automation Manager - Manages analytical work for the instrument based on the Work Order. Produces AWOS’s. Also, manages specimen (automation) work for pre/post processing. Produces SWOS’s
- Analyzer - Instrument that automates IVD testing
- Track - Manages specimen container transport and handoff
- Pre/Post Processor - Device that automates specimen preparation