Laboratory: Difference between revisions
No edit summary |
|||
| Line 112: | Line 112: | ||
== Domain Profile Snapshot == | == Domain Profile Snapshot == | ||
[[Image:LAB_Synopsis2010.jpg| | [[Image:LAB_Synopsis2010.jpg|500px]] | ||
| Line 121: | Line 121: | ||
* [[Laboratory_Point_Of_Care_Testing | LPOCT]] - Laboratory Point Of Care Testing integrates performing and collecting the results of in-vitro testing at the point of care or patient’s bedside. | * [[Laboratory_Point_Of_Care_Testing | LPOCT]] - Laboratory Point Of Care Testing integrates performing and collecting the results of in-vitro testing at the point of care or patient’s bedside. | ||
* [[Laboratory_Code_Sets_Distribution | LCSD]] - Laboratory Code Sets Distribution distributes managed sets of clinical laboratory codes (battery, test and observation codes). | * [[Laboratory_Code_Sets_Distribution | LCSD]] - Laboratory Code Sets Distribution distributes managed sets of clinical laboratory codes (battery, test and observation codes). | ||
== See Also == | == See Also == | ||
Revision as of 13:18, 26 July 2010

IHE Laboratory Domain addresses information sharing and workflow related to in vitro diagnostic testing in clinical laboratories as well as point of care testing. The IHE Laboratory Domain was established in 2003 and manages the Laboratory Profiles and the Laboratory Technical Framework. We are actively soliciting qualified experts from the healthcare profession and healthcare IT industry for ongoing maintenance, new development, and deployment of domain profiles.
The IHE Laboratory Domain is sponsored by:
College of American Pathologists (CAP)
Previous sponsors include:
Groupement pour la Modernisation du Système d'Information Hospitalier (GMSIH)
Société d'Informatique de Laboratoire (SFIL)
- Quick Links
- Laboratory Planning Committee
- Laboratory Technical Committee
- Laboratory Profiles
- Laboratory Technical Framework
- IHE Laboratory Committee Google Group
Timeline : 2010-2011 Planning and Development Cycle
The following table outlines the activities and steps planned for the 2010-2011 year.
The consolidated timeline of all IHE domains can be seen on Domain_Milestone_Dates.
| Timeframe | Activity | Scheduled | Location |
|---|---|---|---|
| 2010 | |||
| Feb 26 - Apr 23 | Planning Committee Call for Proposals | Google Group | |
| May 21 - May 25 | Planning Committee Proposal Vote | Google Group | |
| Aug - Sep | Joint IHE / IICC Calls on ELDA Profile Development | ||
| Sep 30 - Oct 2 | IHE Lab Domain Face to Face Meeting | Chicago, IL USA | |
| 2011 | |||
| May | IHE Lab Domain Face to Face Meeting, Co-chair Elections | TBD | |
| Jun | ELDA Public Comment Period | ||
| Aug | Publication of Trial Implementation ELDA | ||
Current Activity
Next Generation Laboratory Device Automation (LDA)
Demonstrations & Presentations
The following are recent and upcoming public presentations and demonstrations about IHE Lab.
| Date | Location | Event | Details |
|---|---|---|---|
| 2010.07.20 | Webinar | 2010 IHE Webinar Schedule | IHE Education Webinar Series Laboratory Domain |
| 2011.02.20 | Orlando, FL | Interoperability Showcase, HIMSS Annual Conference and Exposition | www.himssconference.org |
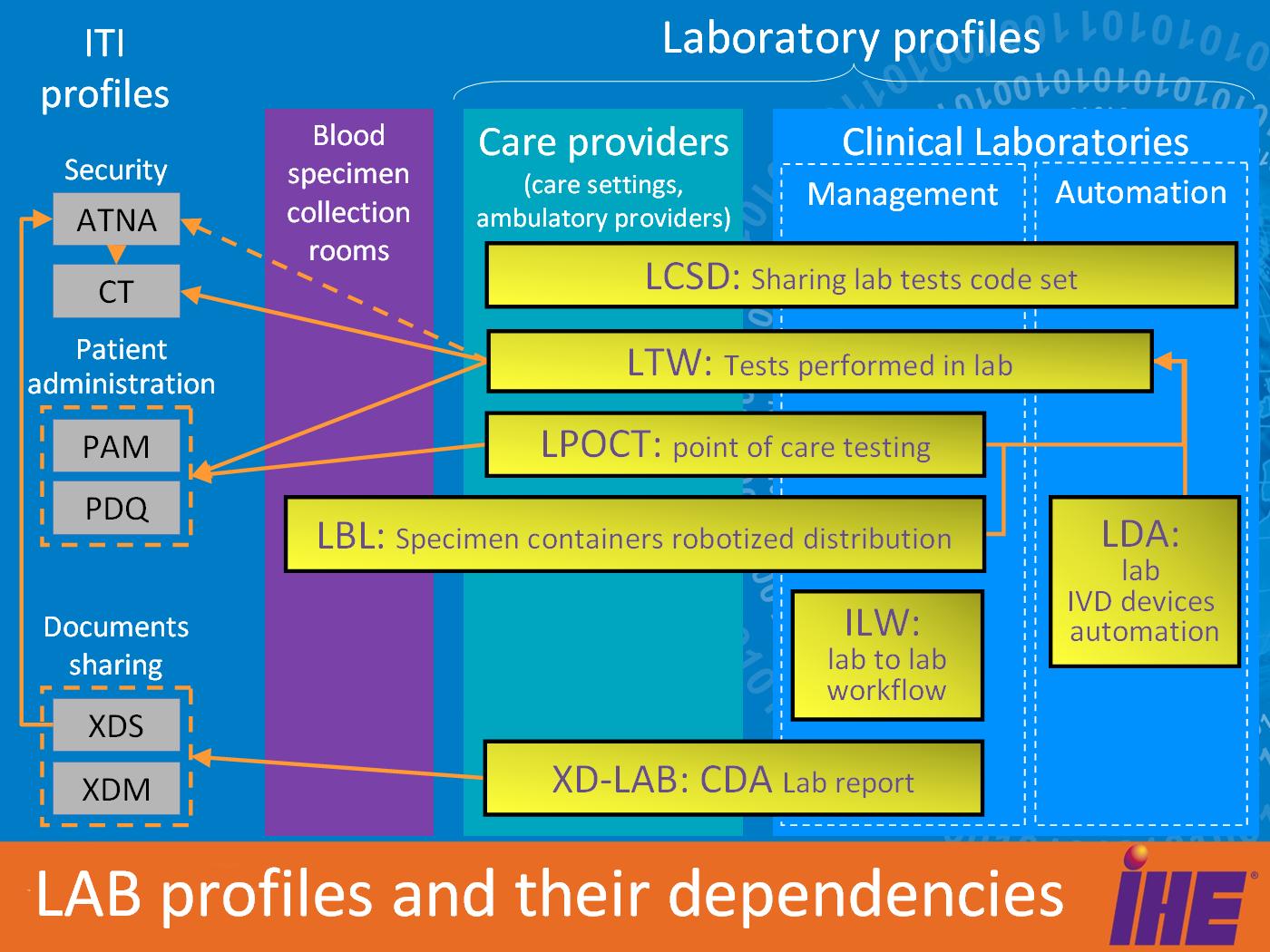
Domain Profile Snapshot
- LTW - Laboratory Testing Workflow integrates ordering and performance of in-vitro diagnostic tests by a clinical laboratory inside a healthcare institution.
- XD-LAB - Sharing Laboratory Reports describes the content (human and machine readable) of an electronic clinical laboratory report.
- LDA - Laboratory Device Automation integrates an Automation Manager and robotic laboratory equipment (pre-analytical devices, analyzers, post-analytical devices) in a clinical lab.
- [[Laboratory_Barcode_Labeling | LBL] - Laboratory Barcode Labeling integrates robotic specimen container labeling systems with sources of order-related labelling information.
- LPOCT - Laboratory Point Of Care Testing integrates performing and collecting the results of in-vitro testing at the point of care or patient’s bedside.
- LCSD - Laboratory Code Sets Distribution distributes managed sets of clinical laboratory codes (battery, test and observation codes).
See Also
This page is based on the Domain Template.