Pathology and Laboratory Medicine (PaLM): Difference between revisions
Riki Merrick (talk | contribs) |
Riki Merrick (talk | contribs) |
||
| Line 45: | Line 45: | ||
* [[PaLM Technical Committee]] | * [[PaLM Technical Committee]] | ||
* [https://docs.google.com/spreadsheets/d/11Ukibo_zURuAFmRA-0p7ZVJ4jUC5ds4c3XXToEzWVFc/edit?usp=sharing PaLM 2024 Meeting Roster] | * [https://docs.google.com/spreadsheets/d/11Ukibo_zURuAFmRA-0p7ZVJ4jUC5ds4c3XXToEzWVFc/edit?usp=sharing PaLM 2024 Meeting Roster] | ||
* [[Profiles# | * [[https://wiki.ihe.net/index.php/Profiles#IHE_Pathology_and_Laboratory_Medicine_Profiles | Published PaLM Profiles]] | ||
* [http://www.ihe.net/Technical_Frameworks/#palm PaLM Technical Framework] | * [http://www.ihe.net/Technical_Frameworks/#palm PaLM Technical Framework] | ||
* [https://groups.google.com/a/ihe.net/forum/#!forum/palm IHE PaLM Google Group] | * [https://groups.google.com/a/ihe.net/forum/#!forum/palm IHE PaLM Google Group] | ||
Revision as of 10:50, 30 October 2025
Origin
The Pathology and Laboratory Medicine (PaLM) domain of IHE becomes active as of January 2016. This domain merges and supersedes the two prior domains Laboratory (LAB) and Anatomic Pathology (AP) respectively launched in 2003 and 2006. The main reason for this merger was the recognition of a significant amount of similarities of the two prior domains scopes, and a long practice of reuse of assets (content modules, transactions) and common thinking from one another. The decision for this merger has been prepared collectively along year 2015 by the LAB and AP leaderships, common secretariat and memberships, and was approved by the Board of IHE International on November 12, 2015.
Scope
The PaLM domain covers:
- the representation and exchange of digital structured data, digital documents, digital images related to ordering, scheduling, performing and reporting diagnostic observations on in-vitro specimens collected from a patient or a non-living subject ;
- the representation and exchange of digital structured data related to specimen management (preparation, transportation, handoff, aliquoting, storage, retrieval)
- the secondary use and exchange of the observation results
- the storage and reuse of diagnostic specimens in bio banks
- transfusion medicine workflows
when a pathology laboratory is involved and in charge of the production of the observation report following the diagnostic/prognostic/screening tests, this laboratory having at least one of the specialties listed below:
| Laboratory specialty | Sub-specialties |
|---|---|
| anatomic pathology specialties | surgical pathology, autopsy, cytopathology, image cytometry, immunohistochemistry |
| clinical pathology specialties | clinical chemistry, hematology, coagulation, blood gas, microbiology, immunology (allergy, auto-immunity, serology), transfusion medicine (blood bank testing), transplant compatibility testing (HLA), fertility, assisted medical procreation, cytogenetic (karyotype, molecular cytogenetic), drug monitoring and toxicology, flow cytometry |
| molecular pathology specialties | gene mutations detection in tumor cells, genetic identification and characterization of infectious agents, diagnostic of genetic disorders |
Sponsorship
The current sponsors of the PaLM domain are:
Prior sponsors of PalM, and the prior domains LAB and/or AP: GMSIH, ADICAP,SEIS, SEAP, SFIL and IHE Japan, ASIP Santé and PHAST.
Current leadership
- Planning co-chairs: Raj Dash, MD - FCAP ; Riki Merrick - Vernetzt
- Technical co-chairs: Alesandro Sulis (CRS4) ; Kenichi Takahashi - Hitachi Hightech
- Secretary and Board representative: Kevin Schap - CAP
Quick links
- PaLM Planning Committee
- PaLM Technical Committee
- PaLM 2024 Meeting Roster
- [| Published PaLM Profiles]
- PaLM Technical Framework
- IHE PaLM Google Group
- PaLM Board Report
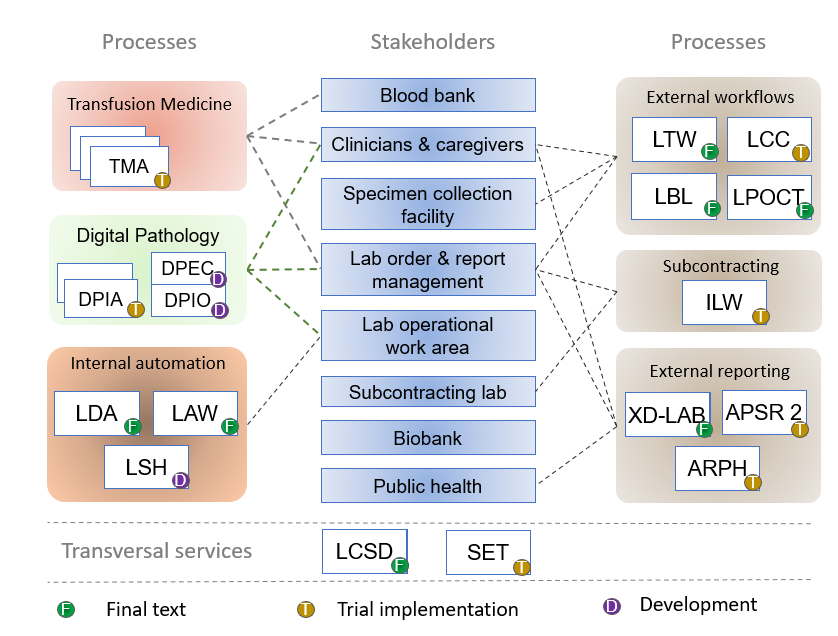
Overview of the PaLM profiles
[Brief descriptions of the published PaLM profiles]
IT Systems that can leverage PaLM profiles
Non-comprehensive list:
- EHR-S in hospital and ambulatory care settings.
- clinical and/or anatomic pathology laboratory information systems (LIS)
- public health information systems and public health lab information systems
- transfusion medicine systems
- electronic healthcare record shared infrastructures (PHR, HIE …)
- robotic specimen container distributers
- barcode labelers
- robotic specimen transportation systems
- robotic peri-analytical devices in the laboratory work area
- IVD analyzers in laboratory or on the point of care
- middleware systems handling a set of analyzers and/or of peri-analytical devices, in laboratory or on the point of care.
- imaging modalities
- PACS and digital archive systems
- biobank management systems
Guidance material and tools
Timeline: 2025 Planning and Development Cycle
Overall publication schedule: See the 2024 development cycle for PaLM
The following table outlines the activities of the domain planned for the 2025 year.
| Timeframe | Activity | Scheduled | Location |
|---|---|---|---|
| Production of new items | |||
| November 3 2024 | Proposals of new work items | Start of period for submission of new item proposals | email to palm@ihe.net |
| December 8 2024 | Proposals of new work items | Closing of period for submission of new item proposals | |
| December 11 2024 | Decision for proposals of new work items finalized on call | ||
| August 2, 2025 | Digital Pathology - Order Workflow (DPOW) | Request for publication of Digital Pathology - Order Workflow (DPOW) Supplement for Public Comment (open for at least 2 months) | email to Mary Jungers |
| October 1, 2025 | Performance Monitoring Workflow (PMW) | Request for publication of Performance Monitoring Workflow (PMW) Supplement for Public Comment (open for at least 2 months) | email to Mary Jungers |
| November 2, 2025 | Digital Pathology - Order Workflow (DPOW) | Request Digital Pathology - Order Workflow (DPOW) Supplement for Trial Implementation | email to Mary Jungers |
| TBD | Technical Framework | Request for publication of Techincal Framework to accommodate Change Requests, if needed | email to Mary Jungers |
| Maintenance of PaLM TF | |||
| Year long | Change proposal process | email to Mary Jungers | |
| Testing | |||
| February 3-7, 2025 | NA Connectathon | https://www.iheusa.org/ihe-north-america-connectathon-week-2025 | Toronto, Canada |
| June 23-27, 2025 | EU Connectathon | https://connectathon.ihe-europe.net/connectathon-week-2025 | Vienna, Austria |
| TBD | Japan Connectathon | UPDATE LINK | UPDATE LOCATION, Japan |
| TBD | Joint DICOM Connectathon | UPDATE LINK | UPDATE LOCATION |
| Meetings | |||
| Year long | Conference calls | 2nd Wednesday of each month 8-10 am (CET), 1st hour: planning & mgmt, 2nd hour: technical | conf calls + Zoom |
| June 23 - 24 | PaLM Committee Face to Face and Joint meeting with DICOM WG 26 at ECDP | Barcelona, Spain | |
| Fall 2025 - date TBD | PaLM Committee Face to face | Location CAP Headquarters, Northfield, IL | |
Current Activity
- Digital Pathology (DP) Profiles - Providing interoperability for solutions in support of a digital pathology workflow.
- IHE PALM's Digital Pathology vision is captured in this article: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8240547/
- Digital Pathology Image Acquisition Profile (DPIA): https://wiki.ihe.net/index.php/Digital_Pathology_Workflow_-_Image_Acquisition - Provides guidance on exchanging data between the Acquisition manager, the scanner and the image manager/archive (PACS)
- Digital Pathology Order Workflow (DPOW) Profile: <add wiki page from template> - Provides guidance for communication of Whole Slide Image related metadata between Image Manager/Archive (PACS) and Order Filler (LIS), also covers communication with Evidence Creator on the order side
- Digital Pathology Evidence Creation (DPEC) Profile: <add wiki page from template> - Providing guidance on reporting AI reuslts of Whole Slide Image analysis by AI (or other systems)
- Digital Pathology Ordering & Reporting (DPOR) Profile: <add wiki page from template> - Providing order and result reporting between AP lab and provider (this is only needed if LTW does not fullfill all AP needs)
- Performance Monitoring Workflow (PMW) Profile: Guidance for exchanging Proficiency Testing results and other performance monitoring data electronically.
- Radiology Pathology Correlation (RPC) Profile - Providing linkages between radiology reports and pathology reports
- [Radiology Pathology Correlation (RPC) profile description|http://wiki.ihe.net/index.php/Radiology_Pathology_Correlation]
- Digital Pathology Aid for Telemedicine (DP-AT) Profile - Sharing digital assests during consultation using telemedicine
- [Digital Pathology Aid for Telemedicine (DP-AT) profile description|http://wiki.ihe.net/index.php/Digital_Pathology_Aid_for_Telemedicine_Profile ]
- Laboratory Specimen Handoff (LSH)
- Specimen Event Tracking (SET) Profile - changes to specimen during processing at all steps from collection, receipt in the lab to storage, including derived material (e.g. slides created from specimen)
- Anatomic Pathology Workflow in an Era of Digital Medicine (APW-EDM):
- Upgrade APW to Digital Pathology i.e. the storage and retrieval of images created from pathology slides - in collaboration with DICOM WG 26
- Digital Pathology initial proposal
- The wiki collaborative space used to prepare the white paper
- Representation of laboratory results in patient summaries using both HL7 CDA and FHIR, LOINC, UCUM and SNOMED CT - collaborations with IHTSDO, Regenstrief and HL7
- Lab result in patient summaries brief proposal
- [http://wiki.hl7.org/index.php?title=International_Patient_Summary_(IPS) International Patient Summary (IPS) project of HL7 International
- IPS CDA R2 IG
- IPS FHIR IG
- International Patient Summary of the Trillium II project funded by Europe EC + US ONC
- Digital Pathology and Structured Reporting (SR) Profile for Clinical Data Capture/Reporting across Multiple Domains
- Current APSR, using CDA standard, is focused on anatomic pathology, so broaden content; this will be a white paper to explore the current state around data element definitions (SDC, DEX) and how these can be integrated into clinical pathways using business rules, without relying on a specific standard for exchange - requires cross domain collaboration (DCC notified)
- SR brief proposal
Published Profile links:
- DPIA
- Laboratory Analytical Workflow (LAW) has reached final text status after a session of intensive testing at the January 2016 NA connectathon. This integration profile, which has been built in a joint project between IHE and the IVD Industry Connectivity Consortium (IICC), will also be published in 2019 as the new standard “AUTO16” for analyzers interfaces by the Clinical Laboratory Standards Institute (CLSI).
- Laboratory Clinical Communication (LCC) profile: standardizing how to indicate suggested replacement orders from the filler side and requesting follow up work by the placer.
- Anatomic Pathology Structured Report release 2.1 (APSR 2.1)
- APSR 2.1 published for Trial Implementation
- ART-DECOR
- example report case 1 (press right click and download)
- example report case 3 (press right click and download)
- example preliminary report version 1 (press right click and download)
- example final report version 2 replacing version 1 (press right click and download)
- the CDA xsl to put in the same folder to view the examples in a web browser (press right click and download)
- IHE Profile to US Realm Lab Guide Gap Analysis
How to Participate
As a Laboratorian, Pathologist, or Advocate
Get involved in the planning committee. This is where the clinical problems are discussed and prioritized. The planning committee is an open group and is interested in having your input. Bring us your interoperability problems. You can also help by participating in the HIMSS showcase scenario definitions.
You can do this by:
- Becoming a IHE Member Organization
- Request to join our IHE PaLM Google Group
- Attend telephone conferences and face to face meetings
- Submit new profile proposals
As a Health IT Implementer
Get involved in the technical committee. Help make sure profiles are feasible and will work for you. Implement the profile, and come experience the IHE Connectathon event. 2011 Connectathon video clip
As a Health IT Buyer
In addition to getting involved in the planning committee, hold your vendors accountable. Put a statement in your RFP that indicates you want the vendor to support the actor in the profile you desire.
Demonstrations & Presentations
The following are recent and upcoming public presentations and demonstrations about IHE Lab. Email our IHE PaLM Google Group about presentations or demonstrations you know about.
| Date | Location | Event | Details |
|---|
Standards Organization - Professional Meetings/Conferences
| Date | Organization | Location | More Information |
|---|---|---|---|
| Feb 10-14, 2025 | HL7 Europe WGM and FHIR Marathon | Lisbon, Portugal | https://www.hl7europe.eu/hl7-working-group-meeting-2025/ |
| Jan 27 - Jan 30, 2025 | HL7 WGM | Virtual on ET | https://www.hl7.org/events/workgroupmeetings.cfm?ref=nav |
| April 6-10, 2025 | SNOMED CT Business Meeting | Oslo, Norway (and online) | https://www.snomed.org/business-meetings |
| May 10-16, 2025 | FHIR Connectathon and HL7 WGM | Madrid, Spain | https://www.hl7.org/events/workgroupmeetings.cfm?ref=nav |
| June 8-12, 2025 | American Society for Clinical Laboratory Science (ASCLS) Annual Meeting | Sacramento, CA | https://ascls.org/annual-meeting/ |
| June 25-28, 2025 | European Congress on Digital Pathology (ECDP 2025) | Barcelona, Spain | https://www.ecdp2025.org/ |
| July 27-31, 2025 | Association for Diagnostics and Laboratody Medicine (ADLM) | Chicago, IL | https://meeting.myadlm.org/ |
| Sep 13-16, 2025 | College of American Pathologist (CAP) Annual Meeting | Orlando, FL | https://events.cap.org/event/ccc369a5-b120-45b3-bc78-b67c8da5a774/home |
| Sep 13-19, 2025 | FHIR Connectathon and HL7 WGM | Pittsburgh, PA | https://www.hl7.org/events/workgroupmeetings.cfm?ref=nav |
| October 5-7, 2025 | PathVisons | San Diego, CA | https://digitalpathologyassociation.org/pathology-visions-conference |
| October 7-10, 2025 | LOINC Conference | Montreal, Canada | https://loinc.org/conference/montreal-2025/ |
| October 18-22, 2025 | SNOMED CT Business Meeting | Antwerp, Belgium (and online) | https://www.snomed.org/business-meetings |
| October 23-25, 2025 | SNOMED CT Expo | Antwerp, Belgium (and online) | https://www.snomed.org/snomedct-expo |
See Also
Milestones for all IHE domains
Standard Organizations Link List
This page is based on the Domain Template.