Specimen Event Tracking
| This profile is part of the Pathology and Laboratory Medicine (PaLM) domain, which merged the former AP and LAB domains since 2016, January 4th. |
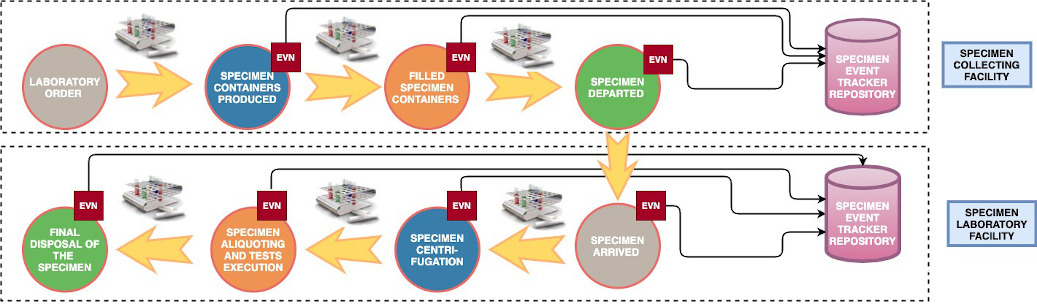
Specimen Event Tracking (SET) profile is a project of the IHE PaLM Technical committee that was started in 2016. The purpose of this new profile of the PaLM domain is to define a series of use cases and all related events relevant to be tracked, when one or more macro operations involving a specimen occur (i.e., container preparation, collection, movement from one facility to another one, archiving, and so on).
Summary
The Specimen Event Tracking (SET) profile covers use cases and transactions related to the tracking of biological specimens in vitro collected for the purpose of diagnostic testing, during their entire lifecycle, from creation to storage inside a laboratory specimen archive or a biobank for future usage, and to final disposal. Specimen workflows can involve a ward and a laboratory in the same hospital, different laboratory facilities inside the same institution, or across different institutions. In the latter case, specimens need to be transferred by a courier service from the sending institution to the receiving institution. Another important use case to be tracked is the specimen creation for the specific purpose of becoming part of a biobank for a research institution or program.
The SET profile tracks macro activities related to specimens, such as collecting, shipping, receiving, accepting... Micro operations part of a macro activity (e.g. decapping a tube) are out of scope of the profile.
For most recent information on the SET please see: http://wiki.ihe.net/index.php/Pathology_and_Laboratory_Medicine_(PaLM)#Current_Activity.
Figure 1 - Some of the main events that might occur to a Specimen during its overall lifecycle, and that the SET profile aims to track. Traceability messages are collected at each step by an Informer entity and sent to a tracker entity (repository). Notice that, according to the use case, the trackers may be more than one, if the Collecting Facility and the Laboratory have independent tracking environments.
Benefits
- Keep under control the chain-of-custody of the specimen, when operation addressed by all the other profiles occur to the specimen
- Prevent specimen loss, opportunity to raise alerts if some exceptions occurs to the process (i.e., broken container, specimen collection unavailable)
- All events might be collected in a single place and use to improve the overall process
Details
Four main use cases have been identified for the SET profile, according to the main operation that might be tracked for a specimen, in different contexts:
- Container Delivery and Specimen Collection Tracking
- Specimen Inter and Intra organization transfer
- Specimen tracking within the Laboratory
- Biobank Specimen Tracking
Each of these use cases describes a scenario where one or more macro-operations might occur to the specimen. For each operation, a correspondent event is tracked by the SET profile. An event is composed of common attributes and specific attributes. For example, all events carry the specimen identifier and the timestamp of occurrence, while only “movement related” events carry information about the departure and the arrival facilities. The SET Profile defines a total of 15 different types of events, which in turn are divided into 4 macro-categories, as shown in the following table:
| SET Events List | |
|---|---|
| Tracking specimen container preparation and specimen collection events | Containers Prepared For Specimen Collection |
| Specimen Collection Succeeded | |
| Specimen Collection Failed | |
| Tracking specimen movements events | Specimen Departed |
| Specimen Arrived | |
| Specimen Accepted | |
| Specimen Rejected | |
| Specimen Archived | |
| Specimen Retrieved from Archive | |
| Specimen Disposed Of | |
| Tracking specimen identification events | Specimen Identifier Changed |
| Tracking specimen processing events | Specimen Procedure Step Successfully Produced a Derived Specimen |
| Specimen Procedure Step Succeeded (with no derived specimen) | |
| Specimen Procedure Step Failed | |
Systems Affected
All systems where an operation to a Specimen occurs are expected to implement this profile.
Actors & Transactions:
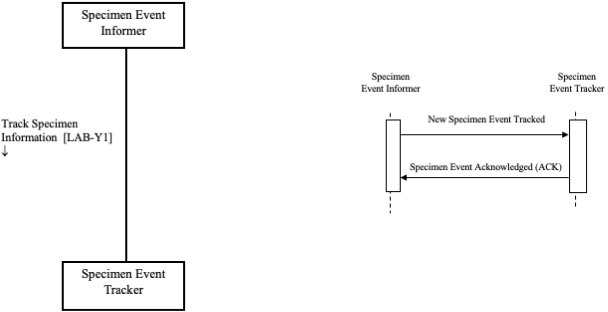
From a point of view of the Actors diagram, a Specimen Event Informer (SEI) actor is defined. This actor is paired with the system where the specimen operation occurs. For example, if the operation is related to the production of the labelled containers for the specimen, the SEI will be paired with a labeling machine.
The Actors and the Interactions diagrams are depicted in Fig. 2:
Figure 2 - Actors (on the left) and Interactions (on the right) diagrams for the SET Profile
Specification
- Profile Status: Trial Implementation
- Documents: SET Supplement
- Important Note: HL7 v2 has been identified as the underlying standard to use. The tracking event information that the HL7v2 message shall carry on strictly depends on the related event type: for example, tracking container preparation for a specimen needs different information than tracking specimen movements or specimen identification. The HL7 v2 messaging standard does not provide a message type that satisfies the needs of all SET event types. For this reason, a CR proposal has been submitted to the HL7 Orders and Observation group, defining a new set of HL7 v2 messages that are specific for Specimen traceability. The last version of the CR proposal is available here