Difference between revisions of "Birth and Fetal Death Reporting Enhanced Profile"
| Line 141: | Line 141: | ||
[[Category:CDA]] | [[Category:CDA]] | ||
[[Category:HL7V2]] | [[Category:HL7V2]] | ||
| + | [[Category:FHIR]] | ||
Revision as of 16:53, 1 June 2018
The Birth and Fetal Death Reporting Extended (BFDR-E) content profile supports pre-population of data from electronic health record systems to electronic vital records systems for birth, fetal death and death reporting to meet Federal, state and local reporting requirements.
Birth and Fetal Death Reporting Enhanced Profile and Vital Records Death Reporting Recording
Summary
The National Vital Statistics System has a long and enduring history that serves to provide essential data on births and deaths within the United States and is the oldest and most successful example of inter-governmental data sharing in Public Health. Currently, these data typically are gathered by hospital personnel from the hospital’s medical records using paper worksheets. The process of capturing Vital Records information manually is duplicative, labor-intensive, costly and can be error prone. As a result, the timeliness and quality of these data are adversely affected.
The IHE Birth and Fetal Death Reporting Extended (BFDR-E) profile defines the mechanism to capture the information required for reporting to vital records. The BFDR-E profile provides the mapping and data capture rules to collect information from the IHE PCC Labor and Delivery Summary using the forms data capture. However, once this information is captured, there is no standard content document or transactional message specified by the BFDR profile to communicate this information to a Vital Records system. This results in limitations in deployment approaches. The recipient of the data must process the custom form data. The Vital Records systems may benefit from the HL7 V2.5.1 Birth and Fetal Death Reporting transaction specification and the HL7 CDA Birth and Fetal Death Reporting document specification to standardize the data they receive.
Benefits
- Establishes interoperable electronic exchange of VR data between EHR and VR Systems
- More timely data release
- Higher quality data for demographic and epidemiologic surveillance and research
- Less costly electronic vital registration systems
- Greater integration with other stakeholder electronic systems
- Greater standardization of electronic Vital Records data collection and exchange
Details
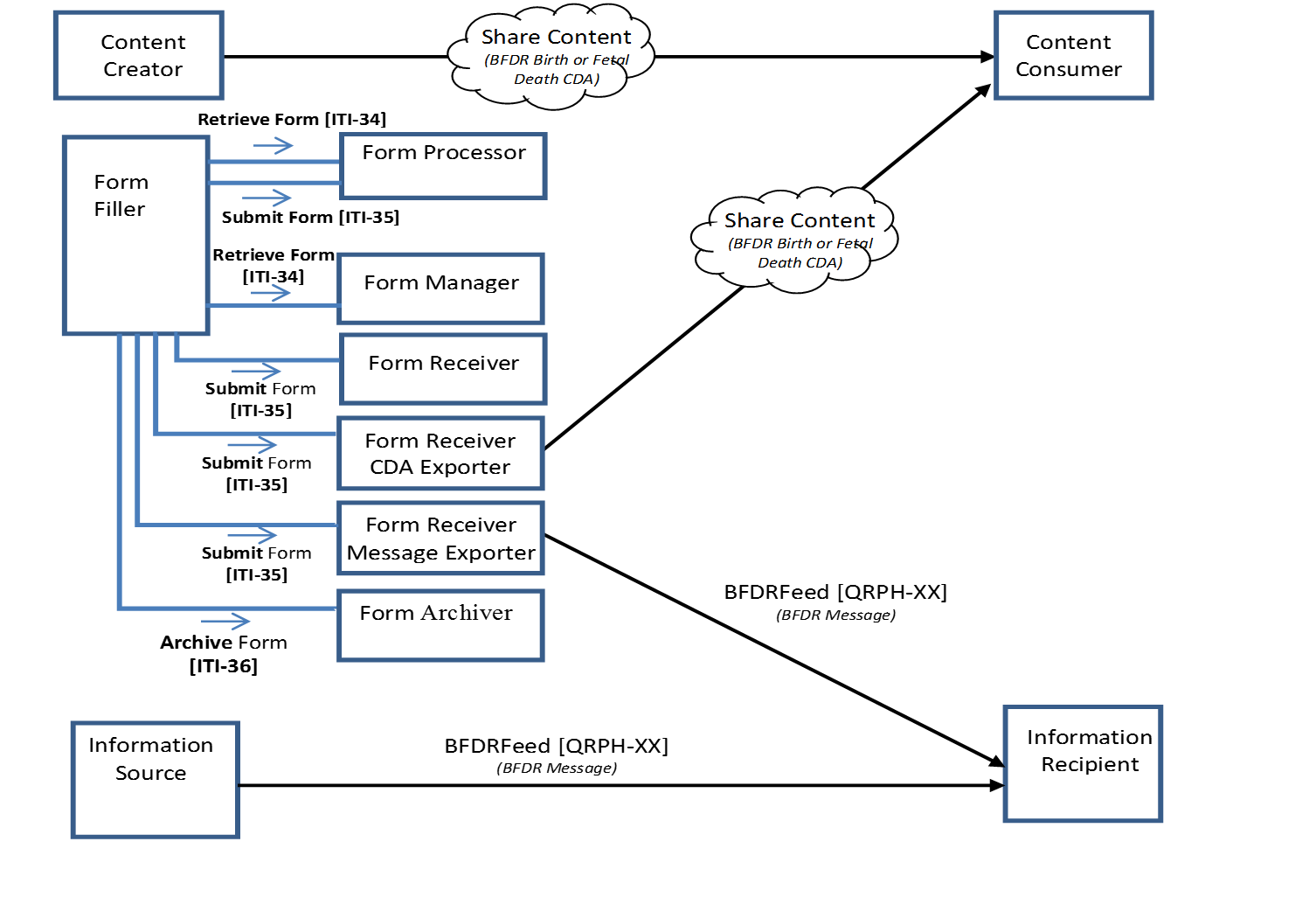
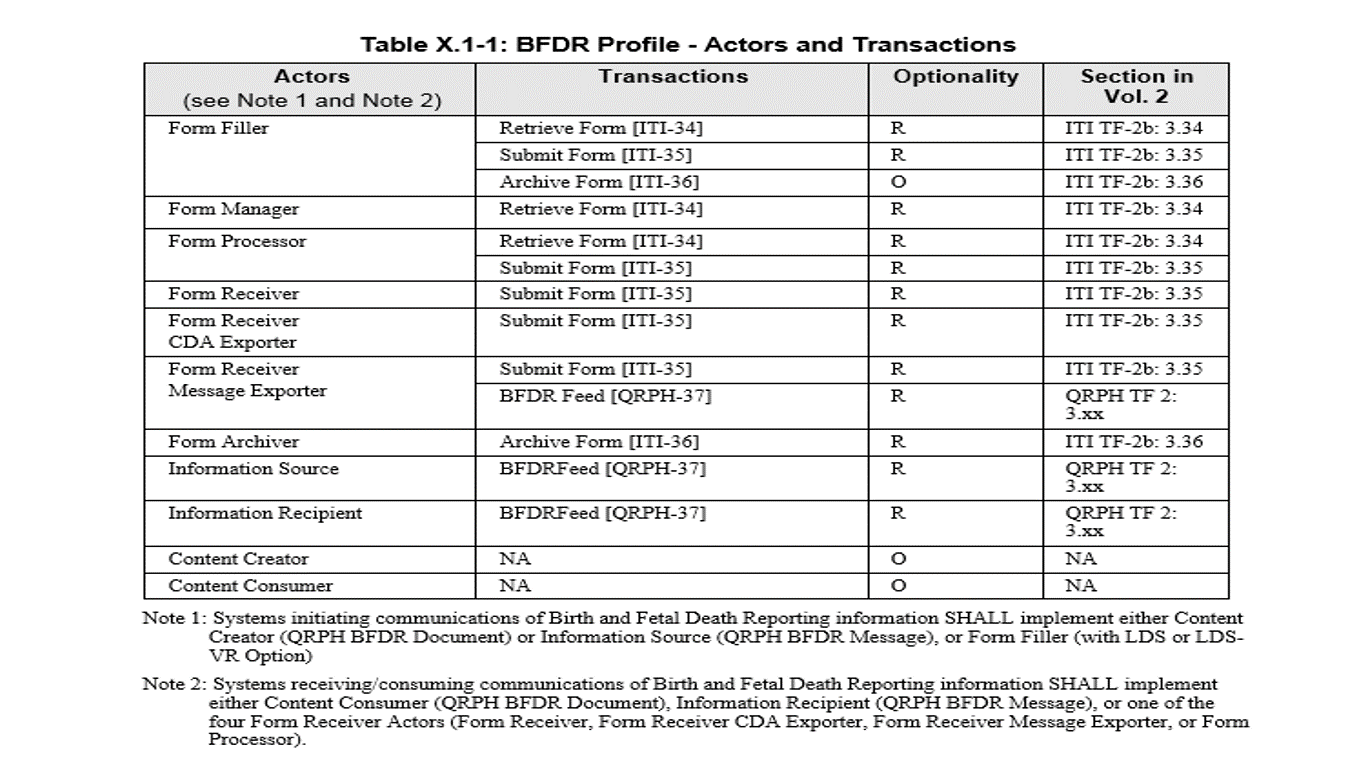
The Birth and Fetal Death Reporting-Enhanced (BFDR-E) Profile provides a means to capture and communicate information needed to report births and fetal deaths for vital registration purposes. BFDR-E builds upon the earlier Birth and Fetal Death Reporting (BFDR) Profile that utilized actors and transactions defined in the ITI Retrieve Form for Data Capture (RFD) Profile to capture structured data using digital forms. BFDR-E further defines a mechanism to transform form submission data and record it in a CDA document designed to exchange the information in a standard format. BFDR-E also defines a mechanism to transform form submission data and transmit it as a standard HL7 v2 message.
Systems Affected
- EHR Systems
- Electronic Birth Registration Systems (EBRD)
- Electronic Fetal Death Registration Systems (EFDRS)
- Infrastructure (Forms Manager)
Specification
Trial Implementation
Documents: http://www.ihe.net/uploadedFiles/Documents/QRPH/IHE_QRPH_Suppl_BFDR-E_Rev1.0_PC_2014-06-06.pdf
Underlying Standards:
•HL7 Version 2.5.1 Implementation Guide: Reporting Birth & Fetal Death Information from the EHR to Vital Records, R1 US Realm (Draft Standard for Trial Use)
•HL7 Version 3 CDA R2 Implementation Guide: Reporting Birth & Fetal Death Information from the EHR to Vital Records Release 1 US Realm - Draft Standard for Trial Use)
FHIR Implementation Guide
Using Resources at FMM Level 2-5:
Composition (FMM 2)
MedicationAdministration (FMM 2)
Procedure (FMM 3)
Observation (FMM 5)
Condition (FMM 3)
Encounter (FMM 2)
Patient (FMM 5)
Coverage (FMM 2)
- The Structure Definitions defined by BFDRE are:
http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.Composition http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MotherMedicationAdministration http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.NewbornMedicationAdministration http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MotherProcedure http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MotherProcedure http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservations which is a composite of: • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsPLUR • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsLIVEB • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsFDTH • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsLLB • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsLMP • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsOPO • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsDOFP • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsPLBD • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsPLBL • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsPPB • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsOWGEST • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsNPCES • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsNPREV • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsPOPO • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsPNC • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsHT • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MaternalObservationsWT
http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.NewbornObservations which is a composite of: • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.NewbornObservationsAPGAR5 • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.NewbornObservationsAPGAR10 • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.NewbornObservationsBW • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.NewbornObservationsFW • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.NewbornObservationsBPLACE • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.NewbornObservationsKaryotype
http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.FetusObservations which is a composite of: • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.FetusObservationsFW • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.FetusObservationsETIME • http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.FetusObservationsHISTOP
http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MotherCondition http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.NewbornCondition http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.FetusCondition http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MotherBirthEncounter http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.NewbornBirthEncounter http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.FetusDeliveryEncounter http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.MotherPatient http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.NewbornPatient http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.FetusPatient http://ihe.net/fhir/StructureDefinition/IHE.BFDRE.Coverage
Informatively this profile will also published on Simplifier. See https://simplifier.net/IHEBFDREimplementation These conformance resources will also be registered at https://registry.fhir.org
- BFDRE ImplementationGuide
- canonical URI http://ihe.net/fhir/ImplementationGuide/IHE.BFDRE
The conformance resources Will also available on the Implementation Material folder.
See ftp://ftp.ihe.net/TF_Implementation_Material/fhir/
See Also
1. HL7 and other standards documents referenced in Volume 1 and Volume 2
2. Birth Edit Specifications for the 2003 Revision of the U.S. Standard Certificate of Live Birth (4/2004; 3/2005; Updated 7/2012)
3. Natality 2003 Revision – File In-Processing Documentation (14 Dec 2010)
4. Fetal Death Edit Specifications for the 2003 Revision of the U.S. Standard Report of Fetal Death