Sharing Laboratory Reports
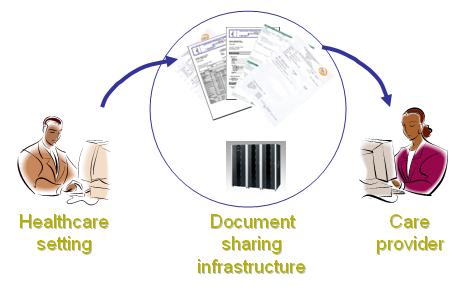
Sharing Laboratory Reports (XD-LAB) describes the laboratory report as an electronic content to be shared among a community of healthcare settings and care providers relying on a document sharing infrastructure.
Summary
This Content Integration Profile describes a clinical laboratory report as an electronic document to be published towards a document sharing resource such as an Electronic Health Record (EHR) or in Personal Health Record (PHR) shared by a community of care providers, using one of the document sharing profiles defined in ITI-TF. Such an electronic document contains the set of releasable results produced by a clinical laboratory in fulfillment of one or more test Orders for a patient. The report is shared in a human-readable format. In addition, this electronic laboratory report SHALL contain test results in a machine-readable format, to facilitate the integration of these observations in the database of a consumer system.
The scope of this profile covers all laboratory specialties except anatomic pathology.
This profile leverages IT Infrastructure profiles [ATNA] and [CT] for security, and [XDS], [XDM], [XDR] for document sharing.
This profile leverages the [Clinical Document Architecture Release 2] (CDAr2) standard from [HL7]
Benefits
Facilitates Patient Care Coordination
- Enables the caregiver to obtain at once the laboratory reports produced during previous episodes of care.
- Reduce over-ordering and over blood sampling from the patient by sharing the last laboratory reports among the various physicians attending the patient.
- Enables the physician to consolidate their patient biological history by importing into their EHR the structured and coded data embedded in the electronic laboratory report.
- Allows a public health laboratory to share broadly its reports related to a suspected outbreak
Details
The Sharing Laboratory Reports (XD-LAB) Integration Profile describes a laboratory report as an electronic document to be shared between various care providers within a document sharing resource. This electronic document has a human-readable format for visualization on screen or print out and embedds each of its reported results as structured and coded data, machine-readable, that can be imported in the database of any consumer of the document.
The XD-LAB Integration Profile covers six major use cases:
- At discharge time, a hospital physician publishing a summary laboratory report with the most significant results obtained during the patient stay.
- The bio-medical scientist of a private laboratory publishes a report for a patient into the regional Patient Health Record.
- An ambulatory physician shares a laboratory report obtained from a laboratory.
- A private or public laboratory publishes automatically all its reports in a shared document repository.
- A healthcare institution produces a cumulative report of all laboratory tests performed for the patient during the encounter.
- A public health laboratory shares its reports into a regional repository.
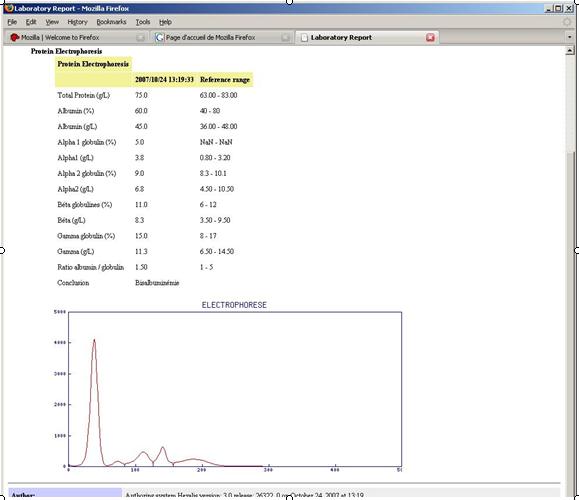
The standard CDAr2 leveraged by XD-LAB enables to share reports embedding images and charts, which are faithfully rendered by a web browser supporting RFC 2397:
Systems Affected
Systems involved in this profile are:
- A system owning a laboratory report and willing to publish it into a document sharing infrastructure. Depending upon the use cases, this can be the system of a clinical laboratory (LIS), the EHR of an ambulatory physician, the EMR of a care setting, the system of a public health laboratory. Each of these systems can play the role of Content Creator.
- The system operated by a professional caregiver who wants to view a laboratory report, and may want to import its structured and coded results into its own database. This system plays the role of Content Consumer.
Actors & Transactions:
Specification
Profile Status: Trial Implementation
Documents:
Underlying Standards:
See Also
Related Profiles
- Integration Profiles XD-LAB depends on
- Cross_Enterprise_Document_Sharing (XDS) or Cross_Enterprise_Document_Media_Interchange (XDM) or Cross_Enterprise_Document_Reliable_Interchange (XDR) from the ITI Technical Framework.
- Audit_Trail_and_Node_Authentication (ATNA) and Consistent_Time (CT) from the ITI Technical Framework.
Consumer Information
This page is based on the Profile Template