Talk:Clinical Research, Public Health and Quality use of EHR Data
Jump to navigation
Jump to search
Presentations from March 14th
Public Health Presentation from Anna Orlova
Orlova-PHDSC%20Presentation%20at%20IHE%203-14-07.ppt Public Health Overview
Quality Presentation from Floyd Eisenberg
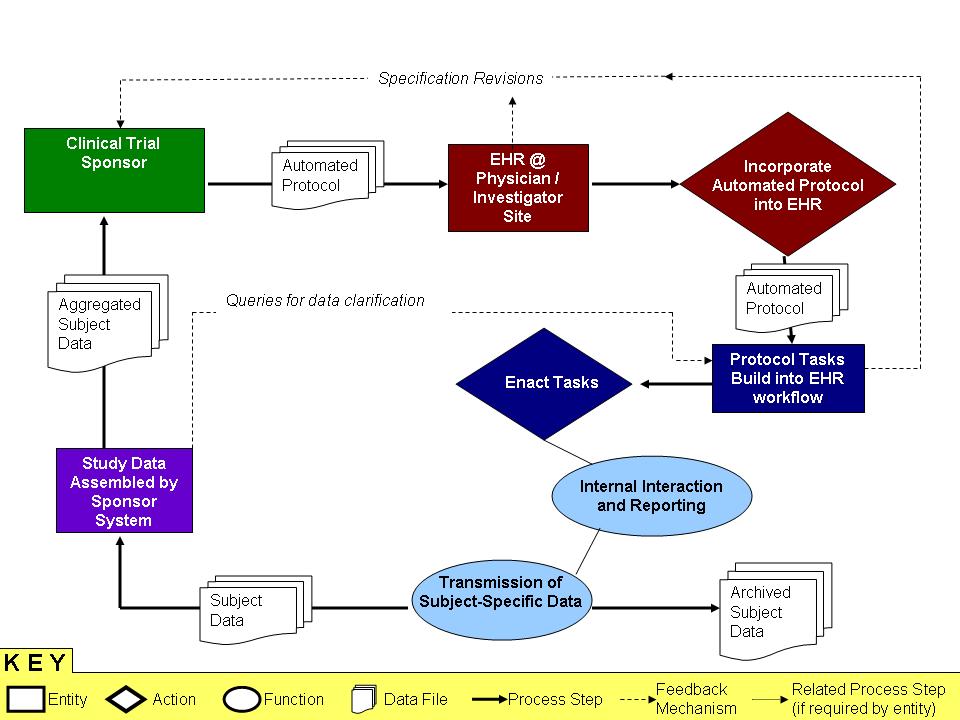
Clinical Trials from Landen Bain
The initial interest of the CDISC community -- the biopharmaceutical companies who sponsor clinical trials -- was to insert a research protocol into an EHR as an executable piece of workflow. While this bears semblance to other 'case management' use cases, the phrase will not make sense to the clinical trial community.
The embedded image (also included as a Powerpoint link) shows how protocol insertion might work.
Media:Media-ProcessDiagramClinicalTrial_CaseMgt_14Mar07_lb.ppt