Radiation Exposure Monitoring for Nuclear Medicine
Radiation Exposure Monitoring for Nuclear Medicine (REM-NM) facilitates the collection and distribution of information about estimated patient radiation exposure resulting from administration of radiopharmaceuticals.
Summary
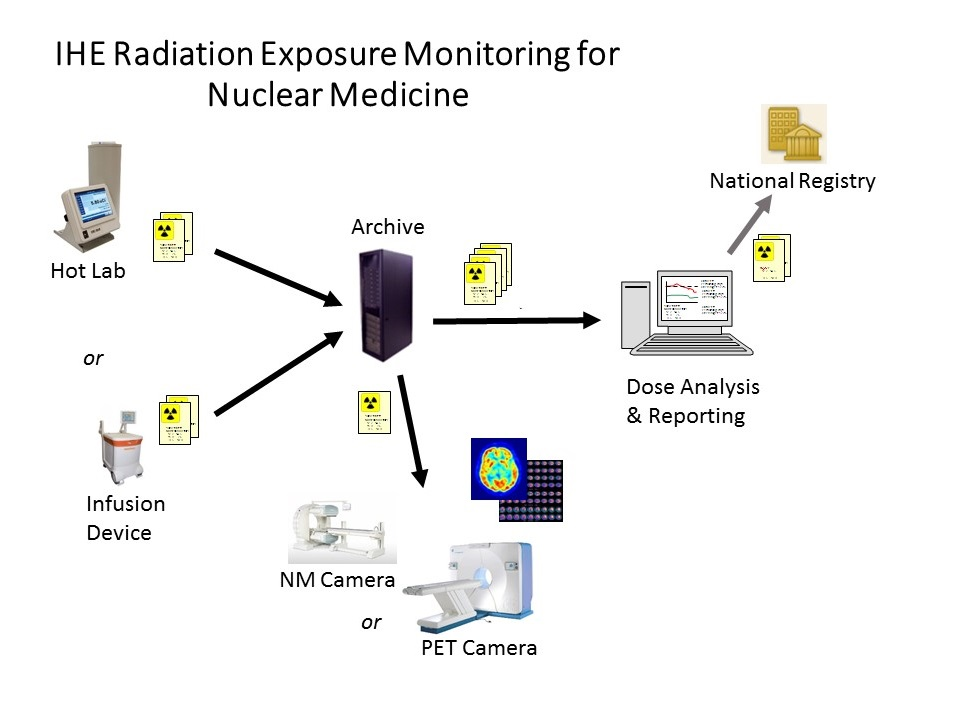
The REM-NM Profile requires radiopharmacutical administration suppliers (e.g. hotlab systems) to export radiopharmaceutical administration details in a standard format and store them to the archive.
The imaging modality is expected to retrieve the dose report, use the details in decay corrections and copy relevant details into the headers of generated images.
The radiation reporting system is expected to perform relevant dose QA analysis and produce related reports. The nature of such analysis and format of the reports is not considered a topic for standardization and is not covered in the profile.
The profile also describes how radiation reporting systems can submit dose reports to centralized registries such as might be run by professional societies or national accreditation groups.
By profiling automated methods, the profile allows dose information to be collected and evaluated without imposing a significant administrative burden on staff otherwise occupied with caring for patients. It is expected that quantitative values such as SUV will be produced more reliably.
Benefits
In the vast majority of medical procedures involving radiation, the potential benefit to the patients’ health from the information gained far outweighs the small potential risk from being exposed to radiation, but the trade-off should not be overlooked. The technological mechanisms provided by this profile can facilitate a conscious evaluation of that trade-off.
REM-NM reports share all the same benefits as REM Reports. Estimating radiation dose delivered to patients for medical purposes can facilitate a number of important activities:
- For facilities exposing patients to radiation, monitoring such exposures can help ensure their policies, procedures and protocols are adequate and being followed appropriately.
- For imaging physicians, monitoring such exposures can assist them in determining how changes in techniques and protocols impact radiation dose as well as image quality. This will enable them to maintain patient doses As Low As Reasonably Achievable (ALARA).
- For patients’ physicians, overall data provided from monitoring such exposures can help them determine (in consultation with the imaging physician) if the benefit from the diagnostic information provided by an individual examination (or additional examinations) outweigh any small risk that may be associated with the imaging exam.
- For medical physicists, having such post-procedure information available for individual patients may help them make essential patient-specific dose estimates for pregnant patients.
- For professional societies and regulatory agencies, a collection of exposure data can be useful when setting or reviewing radiation dose related guidelines. Many such groups have expressed a desire to establish standards of practice or dose reference levels based on a quantitative understanding of current practice, however they have found it prohibitively difficult to collect such data.
- For physicists and physicians, this kind of data can be vital to answering some of the fundamental scientific questions that remain and developing a more detailed understanding of the health impacts of radiation exposure and how it should be measured and managed.
Addition REM-NM benefits
- For PET and NM camera systems, an accurate record of the radiopharmaceutical used, the administered activity, the time of administration and the patient weight can be obtained electronically, reducing data errors and improving the accuracy of PET SUV calculations that depend on that data. Historically, manual user-input errors are common in NM and PET studies.
- Clock synchronization differences between systems that can cause errors in decay estimates (PET radiopharmaceuticals have short half-life’s) and SUV calculations are eliminated by the requirement that both the RAS and the acquisition modalities implement the IHE Consistent Time (CT) Profile.
Details
The REM-NM Profile is based on the work done by DICOM supported by IEC to define DICOM RRD-SR templates (Radiopharmaceutical Radiation Dose SR) appropriate for radiation dose monitoring of NM and PET procedures. These dose objects are created, stored, queried, retrieved, de-identified, and may be processed and displayed just like other DICOM objects such as digital measurements or images. As such, many sites may choose to archive these objects in the study on the PACS together with the images.
The use of DICOM SR objects overcomes Image headers as dose monitoring methods. The SR templates provide far more complete details in a persistent format. If images are deleted or not sent to PACS there are similar gaps in the dose record and Image headers lack important dose details.
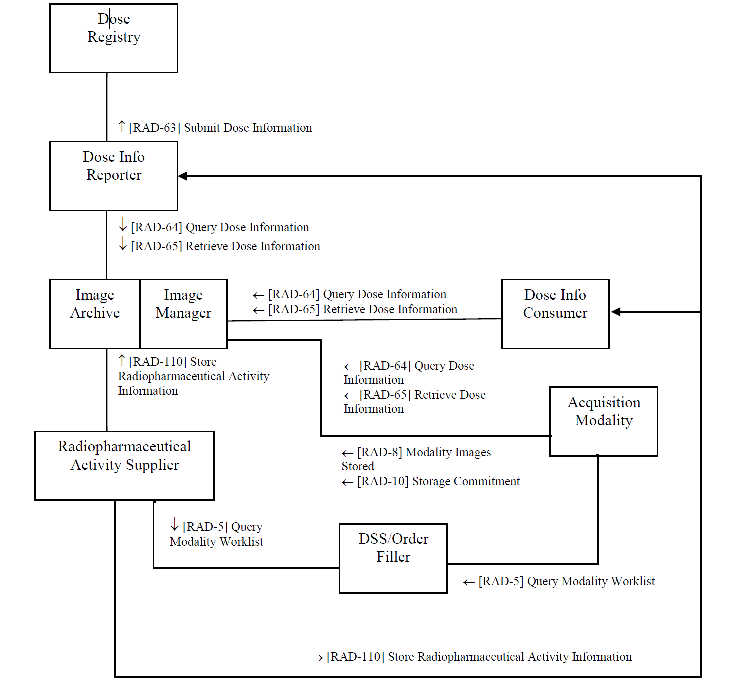
Nuclear Medicine (NM) and Positron Emission Tomography (PET) cameras differ from other modalities in that they do not emit x-ray radiation but rather they detect radiation emitted from a radioactive pharmaceutical (radiopharmaceutical) administered separately to the patients. The administration of the radiopharmaceutical is often performed in a different room minutes before the patient is brought to the NM or PET cameras. Correspondingly the REM-NM report is expected to be originally produced by the radiopharmaceutical administration system (RAS), not the NM or PET camera. The NM or PET camera is expected to access the REM-NM report to populate related fields in the image headers.
Radiopharmaceutical details are recorded for each administration event, which is defined as one radiopharmaceutical administration to a patient. Each administration for a radiopharmaceutical results in one RRD-SR report. The report can describe an administration via a manual injection, infusion from a generator or from an infusion device.
The Profile addresses dose reporting for imaging procedures including Nuclear Medicine, SPECT and Positron Emission Tomography. It does not currently address radiation from implanted seeds.
The exact details recorded in the dose objects may be found in TID 10021 through TID 10024 in DICOM PS 3.16 (see Radiopharmaceutical Radiation Dose).
Some of the key details include:
- Radiopharmaceutical
- Radioisotope
- Half life
- Radiopharmaceutical Administration
- Radiopharmaceutical Administration Event UID
- Radiopharmaceutical Start Date Time
- Administered Activity
- Patient Characteristics
- Patient State, Patient Weight, Patient Weight Time, Glucose
- Activity Information
- Pre-Assay, Post-Assay, value and date time
- Organ Dose Estimate (Optional)
- Organ Name, Organ Dose (mGy), Reference
- Radiopharmaceutical
In addition to supporting quality assurance (QA) of the technical process (was the dose appropriate for the procedure performed) at the local facility, the profile also supports population analysis performed by national registries. Compliant Dose Info Reporter actors are capable of de-identifying and submitting dose reports to a national dose register, making it relatively simple for groups such as ACR to collect and process dose data from across the country once they have recruited participating sites.
While this profile has been strongly supported by medical physicist and radiologist groups, it is important to understand the technical and practical limitations of such dose monitoring and the reasons why the monitored values may not accurately provide the actual radiation dose administered to the patient:
- The values provided by this tool are not “measurements” of dose, but only calculated estimates.
- It is inappropriate and inaccurate to add up dose estimates received by different parts of the body into a single cumulative value.
Despite such limitations, interest in monitoring radiation dose estimates is clearly expressed in such documents as the European directive Euratom 97/43 and the American College of Radiology Dose Whitepaper.
The QIBA FDG-PET committee was also consulted and assisted in developing the standard to support the accurate and consistent dose administration information and patient information that accurate Quantitative Imaging depends on.
Note that the Profile focuses on conveying the details of individual radiopharmaceutical administration events. A proper radiation exposure management program at an imaging facility would involve a medical physicist and define such things as local policies, local reporting requirements, annual reviews, etc. Although this Profile is intended to facilitate such activities, it does not define such policies, reports or processing, or in itself constitute a radiation exposure management program.
Systems Affected
- RAS, Radiopharmaceutical generator, Infusion device and Modalities may create and transmit dose objects.
- PACS systems may store and manage dose objects.
- RIS, PACS or standalone Reporting Workstations may retrieve, process and analyze dose objects.
- Workstations may retrieve, display and possibly supplement dose objects.
- National or regional repositories may receive dose objects submitted by reporting systems at participating sites.
Actors & Transactions:
Specification
Profile Status: Trial Implementation
Documents:
- Radiation Exposure Monitoring for Nuclear Medicine 2016-03-22 (Trial Implementation)
- Current Radiology Technical Framework (includes REM as of Revision 11)
Underlying Standards:
- CPs (that may be relevant, were recently or are not already included in the current DICOM standard and are in varying states of completion) ...
- DICOM CP 1588 Inconsistent Person Participant Relationship in Radiopharmaceutical RDSR
See Also
Related Profiles
- Scheduled Workflow [SWF] can help the modality properly identify the patient in the dose objects.
- Patient Information Reconciliation [PIR] can help the archive update the dose records when patient identifications are updated.
- Portable Data for Imaging [PDI] can store dose objects on media such as CDs.
- Import Reconciliation Workflow [IRWF] can fix patient ids, etc. of dose objects when importing.
- Reporting Workflow [RWF] could conceivably use dose objects as inputs to the reporting process in jurisdictions where dose details are included in the report.
- Simple Image and Numeric Report [SINR] may include data copied from dose objects.
- Cross-enterprise Document Sharing for Imaging [XDS-I] can be used to share dose objects between sites over a network.
- Radiation Exposure Monitoring [REM] specifies how radiation details from imaging procedures are created, exchanged and used.
Presentations
Reference Articles
This page is based on the Profile Template