Data Element Exchange
Data Element Exchange leverages the concept of a metadata registry to add mapping metadata to an annotated data capture form at the point of form design instead of the exchange of data instances.
Data Element Exchange Recording
Summary
To enable clinical research, public health, and quality assessment studies through secondary use of EHR, a mechanism is needed to map EHR data to secondary domain meanings. Integrating patient care and clinical research domains requires a standard-based expressive and scalable interoperability framework for sharing the semantics of the data elements used in patient care and clinical research domains and their mapping to varying data sources. DEX profile enables this through a metadata registry architecture where machine processable definitions of data elements (i.e. metadata) across domains can be shared to address this interoperability challenge to move towards EHR-enabled research. DEX enables retrieving metadata definitions of data elements including “extraction specifications” for a data element defined in a selected domain (like SDTM data elements), from an implementation dependent content model in another domain (like HL7 CCD). As an example, an RFD form Manager can locate the exact mappings of the data elements in the research data set to a pre-population data set provided in HL7 CCD format.
Benefits
- Standardized interaction with a metadata registry to search and retrieve metadata definitions
- Flexible mapping between clinical research and patient care data elements
- Enables dynamic pre-population of clinical research, public health, and quality assessment forms from pre-population data provided by EHR systems as medical summaries
Details
This profile leverages the power of an ISO/IEC 11179 Metadata Registry to search and retrieve data element metadata. The objective is to facilitate creation of annotated data capture form at the point of form design by adding mapping metadata for the data elements in a data capture form to the content models used in clinical care domain. The metadata registry such as CDISC SHARE can serve as the Metadata Source in this profile will define and maintain correspondences between research and healthcare data elements, and will provide an exact map by which an RFD Form Manager can extract data from the pre-population data set.
An electronic data capture system or a research protocol design system would host the Metadata Consumer actor, to query the Metadata Source actor to retrieve the list of data elements hosted and retrieve the metadata of a selected data element including the exact mapping path of the selected data element instance in a content model like HL7 CCD. These mappings would be used to create the annotated data capture forms at the point of form design.
Systems Affected
Metadata Registries may define and maintain metadata of data elements including the correspondences between research and healthcare data elements.
Electronic Data Capture system may query the Metadata Source for a list of Data Elements matching a selection criteria, and may retrieve the metadata of the selected data element.
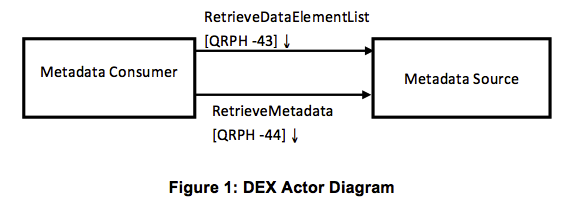
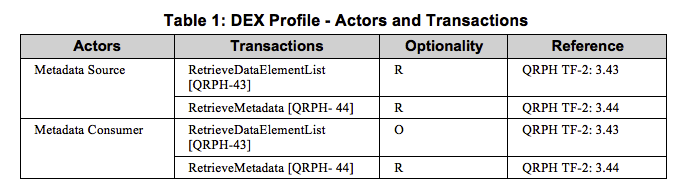
Metadata Consumer The Metadata Consumer is responsible for the importation of metadata created by the Metadata Source. The Metadata Consumer can optionally query the Metadata Source for a list of Data Elements matching a selection criteria.
Metadata Source The Metadata Source is responsible for creation of the Data Element list matching a selection criteria and the creation of metadata for a selected Data Element per request from the Metadata Consumer. The Metadata Source is associated with a metadata registry.
Specification
Profile Status: Profile Status: Trial Implementation
Documents:
Data Element Exchange (DEX) -Supplement for Trial Implementation- Published 2013-09-13
Underlying Standards:
- ISO/IEC 11179: Information Technology – Metadata Registries (MDR) Parts 1–6 (3rd Edition).
- IETF RFC2616 HyperText Transfer Protocol HTTP/1.1
- Extensible Markup Language (XML) 1.0 (Second Edition). W3C Recommendation 6 October 2000. http://www.w3.org/TR/REC-xml.
- Web Services Description Language (WSDL) 1.1. W3C Note 15 March 2001. http://www.w3.org/TR/wsdl.
- SOAP 1.2 Second Edition, W3C Recommendation 27 April 2007. http://www.w3.org/TR/soap12-part1
- IEEE Std 1003.2 IEEE Standard for Information Technology — Portable Operating System Interface (POSIX®) — Part 2: Shell and Utilities — Amendment 1: Batch Environment -Description